A review of heterogeneous photocatalysis for water and surface disinfection
- PMID: 25830789
- PMCID: PMC6272584
- DOI: 10.3390/molecules20045574
A review of heterogeneous photocatalysis for water and surface disinfection
Abstract
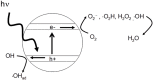
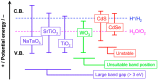
Photo-excitation of certain semiconductors can lead to the production of reactive oxygen species that can inactivate microorganisms. The mechanisms involved are reviewed, along with two important applications. The first is the use of photocatalysis to enhance the solar disinfection of water. It is estimated that 750 million people do not have accessed to an improved source for drinking and many more rely on sources that are not safe. If one can utilize photocatalysis to enhance the solar disinfection of water and provide an inexpensive, simple method of water disinfection, then it could help reduce the risk of waterborne disease. The second application is the use of photocatalytic coatings to combat healthcare associated infections. Two challenges are considered, i.e., the use of photocatalytic coatings to give "self-disinfecting" surfaces to reduce the risk of transmission of infection via environmental surfaces, and the use of photocatalytic coatings for the decontamination and disinfection of medical devices. In the final section, the development of novel photocatalytic materials for use in disinfection applications is reviewed, taking account of materials, developed for other photocatalytic applications, but which may be transferable for disinfection purposes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- IUPAC . Compendium of Chemical Terminology. In: McNaught A.D., Wilkinson A., editors. The Gold Book. 2nd ed. Blackwell Scientific Publications; Oxford, UK: 1997.
-
- Mills A., Le Hunte S. An overview of semiconductor photocatalysis. J. Photochem. Photobiol. A. 1997;108:1–35. doi: 10.1016/S1010-6030(97)00118-4. - DOI
-
- Tryk D.A., Fujishima A., Honda K. Recent topics in photoelectrochemistry: Achievements and future prospects. Electrochim. Acta. 2000;45:2363–2376. doi: 10.1016/S0013-4686(00)00337-6. - DOI
-
- Bahnemann D. Photocatalytic water treatment: Solar energy applications. Sol. Energy. 2004;77:445–459. doi: 10.1016/j.solener.2004.03.031. - DOI
-
- Agrios A.G., Pichat P. State of the art and perspectives on materials and applications of photocatalysis over TiO2. J. Appl. Electrochem. 2005;35:655–663. doi: 10.1007/s10800-005-1627-6. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

