Affinity chromatography of the bovine cerebral cortex A1 adenosine receptor
- PMID: 2583275
- PMCID: PMC5549270
- DOI: 10.1016/0014-5793(89)81555-8
Affinity chromatography of the bovine cerebral cortex A1 adenosine receptor
Abstract
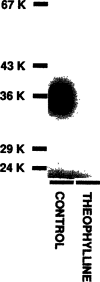
An approximate 140-fold purification of the A1 adenosine receptor of bovine cerebral cortex has been obtained via affinity chromatography. The affinity column consists of Affi-Gel 10 coupled through an amide linkage to XAC, a high-affinity A1 adenosine receptor antagonist. As assessed by [3H]XAC binding, bovine brain membranes solubilized with the detergent CHAPS had a specific binding activity of 1.1 pmol/mg protein. Interaction of solubilized A1 adenosine receptors with the XAC-Affi-Gel was biospecific and 30% of the receptor activity was bound by the gel. Demonstration of [3H]XAC binding in the material eluted from the column with R-PIA required insertion of receptor into phospholipid vesicles. The specific activity of the affinity column purified receptor was 146 +/- 22 pmol/mg protein with typically 5-15% of the bound receptor recovered. The purified receptor displayed high-affinity antagonist binding and bound agonists with the potency order expected of the bovine brain A1 adenosine receptor: R-PIA greater than S-PIA greater than NECA. In purified preparations, the photoaffinity probe [125I]PAPAXAC-SANPAH specifically labelled a protein of molecular mass 38,000 which has previously been shown to be the A1 adenosine receptor binding subunit.
Figures
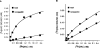
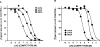
Similar articles
-
Purification and characterization of bovine cerebral cortex A1 adenosine receptor.Arch Biochem Biophys. 1990 Dec;283(2):440-6. doi: 10.1016/0003-9861(90)90665-l. Arch Biochem Biophys. 1990. PMID: 2275555 Free PMC article.
-
Affinity chromatography of A1 adenosine receptors of rat brain membranes.Mol Pharmacol. 1989 Jun;35(6):780-6. Mol Pharmacol. 1989. PMID: 2733695
-
Demonstration of both A1 and A2 adenosine receptors in DDT1 MF-2 smooth muscle cells.Mol Pharmacol. 1990 Feb;37(2):149-56. Mol Pharmacol. 1990. PMID: 2304450 Free PMC article.
-
Co-purification of A1 adenosine receptors and guanine nucleotide-binding proteins from bovine brain.J Biol Chem. 1989 Sep 5;264(25):14853-9. J Biol Chem. 1989. PMID: 2504712
-
A new high affinity, iodinated adenosine receptor antagonist as a radioligand/photoaffinity crosslinking probe.Mol Pharmacol. 1987 Aug;32(1):184-8. Mol Pharmacol. 1987. PMID: 3614192 Free PMC article.
Cited by
-
Adenosine A1 and A2 receptors: structure--function relationships.Med Res Rev. 1992 Sep;12(5):423-71. doi: 10.1002/med.2610120502. Med Res Rev. 1992. PMID: 1513184 Free PMC article. Review. No abstract available.
-
Extracellular ATP stimulates three different receptor-signal transduction systems in FRTL-5 thyroid cells. Activation of phospholipase C, and inhibition and activation of adenylate cyclase.Biochem J. 1992 Apr 1;283 ( Pt 1)(Pt 1):281-7. doi: 10.1042/bj2830281. Biochem J. 1992. PMID: 1314567 Free PMC article.
-
Identification of an adenosine receptor domain specifically involved in binding of 5'-substituted adenosine agonists.J Biol Chem. 1994 Jul 8;269(27):18016-20. J Biol Chem. 1994. PMID: 8027060 Free PMC article.
-
Xanthines as adenosine receptor antagonists.Handb Exp Pharmacol. 2011;(200):151-99. doi: 10.1007/978-3-642-13443-2_6. Handb Exp Pharmacol. 2011. PMID: 20859796 Free PMC article. Review.
-
[(3)H]XAC (xanthine amine congener) is a radioligand for A(2)-adenosine receptors in rabbit striatum.Neurochem Int. 1991;18(2):207-13. doi: 10.1016/0197-0186(91)90187-i. Neurochem Int. 1991. PMID: 20504695 Free PMC article.
References
-
- Ramkumar V, Pierson G, Stiles GL. In: Progress in Drug Research. Jucker E, editor. Vol. 32. Birkhauer, Basel; 1988. pp. 195–247. - PubMed
-
- Williams M. Annu. Rev. Pharmacol. Toxicol. 1987;27:315–345. - PubMed
-
- Stiles GL. J. Biol. Chem. 1986;261:10839–10843. - PubMed
-
- Gavish M, Goodman RR, Snyder SH. Science. 1983;215:1633–1635. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

