Novel cross talk between IGF-IR and DDR1 regulates IGF-IR trafficking, signaling and biological responses
- PMID: 25840417
- PMCID: PMC4599258
- DOI: 10.18632/oncotarget.3177
Novel cross talk between IGF-IR and DDR1 regulates IGF-IR trafficking, signaling and biological responses
Abstract
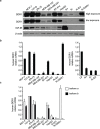
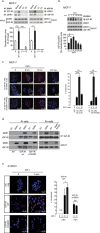
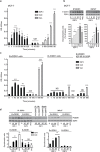
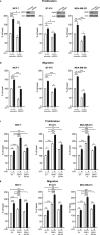
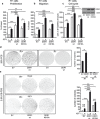
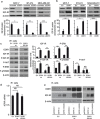
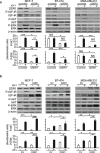
The insulin-like growth factor-I receptor (IGF-IR), plays a key role in regulating mammalian development and growth, and is frequently deregulated in cancer contributing to tumor initiation and progression. Discoidin domain receptor 1 (DDR1), a collagen receptor tyrosine-kinase, is as well frequently overexpressed in cancer and implicated in cancer progression. Thus, we investigated whether a functional cross-talk between the IGF-IR and DDR1 exists and plays any role in cancer progression.Using human breast cancer cells we found that DDR1 constitutively associated with the IGF-IR. However, this interaction was enhanced by IGF-I stimulation, which promoted rapid DDR1 tyrosine-phosphorylation and co-internalization with the IGF-IR. Significantly, DDR1 was critical for IGF-IR endocytosis and trafficking into early endosomes, IGF-IR protein expression and IGF-I intracellular signaling and biological effects, including cell proliferation, migration and colony formation. These biological responses were inhibited by DDR1 silencing and enhanced by DDR1 overexpression.Experiments in mouse fibroblasts co-transfected with the human IGF-IR and DDR1 gave similar results and indicated that, in the absence of IGF-IR, collagen-dependent phosphorylation of DDR1 is impaired.These results demonstrate a critical role of DDR1 in the regulation of IGF-IR action, and identify DDR1 as a novel important target for breast cancers that overexpress IGF-IR.
Keywords: DDR1; IGF-IR; breast cancer; insulin-like growth factor-I receptor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
IGF-I induces upregulation of DDR1 collagen receptor in breast cancer cells by suppressing MIR-199a-5p through the PI3K/AKT pathway.Oncotarget. 2016 Feb 16;7(7):7683-700. doi: 10.18632/oncotarget.6524. Oncotarget. 2016. PMID: 26655502 Free PMC article.
-
Discoidin domain receptor 1 modulates insulin receptor signaling and biological responses in breast cancer cells.Oncotarget. 2017 Jun 27;8(26):43248-43270. doi: 10.18632/oncotarget.18020. Oncotarget. 2017. PMID: 28591735 Free PMC article.
-
Collagen stimulates discoidin domain receptor 1-mediated migration of smooth muscle cells through Src.Cardiovasc Pathol. 2011 Mar-Apr;20(2):71-6. doi: 10.1016/j.carpath.2009.12.006. Epub 2010 Jan 25. Cardiovasc Pathol. 2011. PMID: 20093046
-
A novel functional crosstalk between DDR1 and the IGF axis and its relevance for breast cancer.Cell Adh Migr. 2018;12(4):305-314. doi: 10.1080/19336918.2018.1445953. Epub 2018 Mar 23. Cell Adh Migr. 2018. PMID: 29486622 Free PMC article. Review.
-
The Emerging Role of Insulin Receptor Isoforms in Thyroid Cancer: Clinical Implications and New Perspectives.Int J Mol Sci. 2018 Nov 30;19(12):3814. doi: 10.3390/ijms19123814. Int J Mol Sci. 2018. PMID: 30513575 Free PMC article. Review.
Cited by
-
RAGE inhibition blunts insulin-induced oncogenic signals in breast cancer.Breast Cancer Res. 2023 Jul 17;25(1):84. doi: 10.1186/s13058-023-01686-5. Breast Cancer Res. 2023. PMID: 37461077 Free PMC article.
-
Cabozantinib Is Effective in Melanoma Brain Metastasis Cell Lines and Affects Key Signaling Pathways.Int J Mol Sci. 2021 Nov 14;22(22):12296. doi: 10.3390/ijms222212296. Int J Mol Sci. 2021. PMID: 34830178 Free PMC article.
-
Progranulin/EphA2 axis: A novel oncogenic mechanism in bladder cancer.Matrix Biol. 2020 Nov;93:10-24. doi: 10.1016/j.matbio.2020.03.009. Epub 2020 May 15. Matrix Biol. 2020. PMID: 32417448 Free PMC article.
-
IGF-I induces upregulation of DDR1 collagen receptor in breast cancer cells by suppressing MIR-199a-5p through the PI3K/AKT pathway.Oncotarget. 2016 Feb 16;7(7):7683-700. doi: 10.18632/oncotarget.6524. Oncotarget. 2016. PMID: 26655502 Free PMC article.
-
LRP-1 Promotes Colon Cancer Cell Proliferation in 3D Collagen Matrices by Mediating DDR1 Endocytosis.Front Cell Dev Biol. 2020 Jun 3;8:412. doi: 10.3389/fcell.2020.00412. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32582700 Free PMC article.
References
-
- Brogiolo W, Stocker H, Ikeya T, Rintelen F, Fernandez R, Hafen E. An evolutionarily conserved function of the Drosophila insulin receptor and insulin-like peptides in growth control. Curr Biol. 2001;11:213–221. - PubMed
-
- Stewart CE, Rotwein P. Growth, differentiation, and survival: multiple physiological functions for insulin-like growth factors. Physiol Rev. 1996;76:1005–1026. - PubMed
-
- Bartke A. Minireview: role of the growth hormone/insulin-like growth factor system in mammalian aging. Endocrinology. 2005;146:3718–3723. - PubMed
-
- Belfiore A, Frittitta L, Costantino A, Frasca F, Pandini G, Sciacca L, et al. Insulin receptors in breast cancer. Ann N Y Acad Sci. 1996;784:173–188. - PubMed
-
- Carboni JM, Lee A V, Hadsell DL, Rowley BR, Lee FY, Bol DK, et al. Tumor development by transgenic expression of a constitutively active insulin-like growth factor I receptor. Cancer Res. 2005;65:3781–3787. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

