Tetraspanin CD9 participates in dysmegakaryopoiesis and stromal interactions in primary myelofibrosis
- PMID: 25840601
- PMCID: PMC4450621
- DOI: 10.3324/haematol.2014.118497
Tetraspanin CD9 participates in dysmegakaryopoiesis and stromal interactions in primary myelofibrosis
Abstract
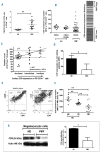
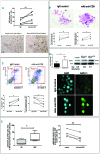
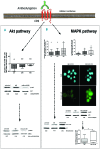
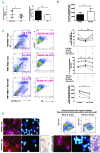
Primary myelofibrosis is characterized by clonal myeloproliferation, dysmegakaryopoiesis, extramedullary hematopoiesis associated with myelofibrosis and altered stroma in the bone marrow and spleen. The expression of CD9, a tetraspanin known to participate in megakaryopoiesis, platelet formation, cell migration and interaction with stroma, is deregulated in patients with primary myelofibrosis and is correlated with stage of myelofibrosis. We investigated whether CD9 participates in the dysmegakaryopoiesis observed in patients and whether it is involved in the altered interplay between megakaryocytes and stromal cells. We found that CD9 expression was modulated during megakaryocyte differentiation in primary myelofibrosis and that cell surface CD9 engagement by antibody ligation improved the dysmegakaryopoiesis by restoring the balance of MAPK and PI3K signaling. When co-cultured on bone marrow mesenchymal stromal cells from patients, megakaryocytes from patients with primary myelofibrosis displayed modified behaviors in terms of adhesion, cell survival and proliferation as compared to megakaryocytes from healthy donors. These modifications were reversed after antibody ligation of cell surface CD9, suggesting the participation of CD9 in the abnormal interplay between primary myelofibrosis megakaryocytes and stroma. Furthermore, silencing of CD9 reduced CXCL12 and CXCR4 expression in primary myelofibrosis megakaryocytes as well as their CXCL12-dependent migration. Collectively, our results indicate that CD9 plays a role in the dysmegakaryopoiesis that occurs in primary myelofibrosis and affects interactions between megakaryocytes and bone marrow stromal cells. These results strengthen the "bad seed in bad soil" hypothesis that we have previously proposed, in which alterations of reciprocal interactions between hematopoietic and stromal cells participate in the pathogenesis of primary myelofibrosis.
Copyright© Ferrata Storti Foundation.
Figures
Similar articles
-
FLT3-mediated p38-MAPK activation participates in the control of megakaryopoiesis in primary myelofibrosis.Cancer Res. 2011 Apr 15;71(8):2901-15. doi: 10.1158/0008-5472.CAN-10-1731. Epub 2011 Apr 12. Cancer Res. 2011. PMID: 21487043
-
Difference in megakaryocyte expression of GATA-1, IL-6, and IL-8 associated with maintenance of platelet counts in patients with plasma cell neoplasm with dysmegakaryopoiesis.Exp Hematol. 2019 May;73:13-17.e2. doi: 10.1016/j.exphem.2019.02.005. Epub 2019 Feb 27. Exp Hematol. 2019. PMID: 30825517 Clinical Trial.
-
Downregulation of GATA1 drives impaired hematopoiesis in primary myelofibrosis.J Clin Invest. 2017 Apr 3;127(4):1316-1320. doi: 10.1172/JCI82905. Epub 2017 Feb 27. J Clin Invest. 2017. PMID: 28240607 Free PMC article.
-
Pathogenesis of myelofibrosis: role of ineffective megakaryopoiesis and megakaryocyte components.Prog Clin Biol Res. 1984;154:427-54. Prog Clin Biol Res. 1984. PMID: 6089232 Review.
-
Megakaryocyte-bone cell interactions: lessons from mouse models of experimental myelofibrosis and related disorders.Am J Physiol Cell Physiol. 2022 Feb 1;322(2):C177-C184. doi: 10.1152/ajpcell.00328.2021. Epub 2021 Dec 15. Am J Physiol Cell Physiol. 2022. PMID: 34910601 Review.
Cited by
-
Patients with Waldenström macroglobulinemia have impaired platelet and coagulation function.Blood Adv. 2024 Nov 12;8(21):5542-5555. doi: 10.1182/bloodadvances.2024014190. Blood Adv. 2024. PMID: 39207869 Free PMC article.
-
Tspan8 and Tspan8/CD151 knockout mice unravel the contribution of tumor and host exosomes to tumor progression.J Exp Clin Cancer Res. 2018 Dec 12;37(1):312. doi: 10.1186/s13046-018-0961-6. J Exp Clin Cancer Res. 2018. Retraction in: J Exp Clin Cancer Res. 2025 Jan 16;44(1):16. doi: 10.1186/s13046-025-03284-z. PMID: 30541597 Free PMC article. Retracted.
-
Transcriptome analysis of bone marrow mesenchymal stromal cells from patients with primary myelofibrosis.Genom Data. 2015 Apr 29;5:1-2. doi: 10.1016/j.gdata.2015.04.017. eCollection 2015 Sep. Genom Data. 2015. PMID: 26484208 Free PMC article.
-
Dynamins 2 and 3 control the migration of human megakaryocytes by regulating CXCR4 surface expression and ITGB1 activity.Blood Adv. 2018 Dec 11;2(23):3540-3552. doi: 10.1182/bloodadvances.2018021923. Blood Adv. 2018. PMID: 30538113 Free PMC article.
-
Differential Expression of the Tetraspanin CD9 in Normal and Leukemic Stem Cells.Biology (Basel). 2021 Apr 8;10(4):312. doi: 10.3390/biology10040312. Biology (Basel). 2021. PMID: 33918035 Free PMC article.
References
-
- Lataillade JJ, Pierre-Louis O, Hasselbalch HC, et al. Does primary myelofibrosis involve a defective stem cell niche? From concept to evidence. Blood. 2008;112(8): 3026–3035. - PubMed
-
- James C, Ugo V, Le Couedic JP, Staerk J, et al. A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature. 2005;434(7037):1144–1148. - PubMed
-
- Pardanani AD, Levine RL, Lasho T, et al. MPL515 mutations in myeloproliferative and other myeloid disorders: a study of 1182 patients. Blood. 2006;108(10):3472–3476. - PubMed
-
- Stegelmann F, Bullinger L, Schlenk RF, et al. DNMT3A mutations in myeloproliferative neoplasms. Leukemia. 2011;25(7):1217–1219. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

