Plasma membrane mechanical stress activates TRPC5 channels
- PMID: 25849346
- PMCID: PMC4388645
- DOI: 10.1371/journal.pone.0122227
Plasma membrane mechanical stress activates TRPC5 channels
Abstract
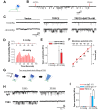
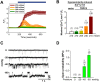
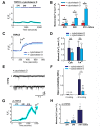
Mechanical forces exerted on cells impose stress on the plasma membrane. Cells sense this stress and elicit a mechanoelectric transduction cascade that initiates compensatory mechanisms. Mechanosensitive ion channels in the plasma membrane are responsible for transducing the mechanical signals to electrical signals. However, the mechanisms underlying channel activation in response to mechanical stress remain incompletely understood. Transient Receptor Potential (TRP) channels serve essential functions in several sensory modalities. These channels can also participate in mechanotransduction by either being autonomously sensitive to mechanical perturbation or by coupling to other mechanosensory components of the cell. Here, we investigated the response of a TRP family member, TRPC5, to mechanical stress. Hypoosmolarity triggers Ca2+ influx and cationic conductance through TRPC5. Importantly, for the first time we were able to record the stretch-activated TRPC5 current at single-channel level. The activation threshold for TRPC5 was found to be 240 mOsm for hypoosmotic stress and between -20 and -40 mmHg for pressure applied to membrane patch. In addition, we found that disruption of actin filaments suppresses TRPC5 response to hypoosmotic stress and patch pipette pressure, but does not prevent the activation of TRPC5 by stretch-independent mechanisms, indicating that actin cytoskeleton is an essential transduction component that confers mechanosensitivity to TRPC5. In summary, our findings establish that TRPC5 can be activated at the single-channel level when mechanical stress on the cell reaches a certain threshold.
Conflict of interest statement
Figures

Similar articles
-
Hypoosmotic- and pressure-induced membrane stretch activate TRPC5 channels.J Physiol. 2008 Dec 1;586(23):5633-49. doi: 10.1113/jphysiol.2008.161257. Epub 2008 Oct 2. J Physiol. 2008. PMID: 18832422 Free PMC article.
-
G protein-coupled receptor signalling potentiates the osmo-mechanical activation of TRPC5 channels.Pflugers Arch. 2014 Aug;466(8):1635-46. doi: 10.1007/s00424-013-1392-z. Epub 2013 Nov 1. Pflugers Arch. 2014. PMID: 24177920
-
Ca2+-calmodulin-dependent myosin light chain kinase is essential for activation of TRPC5 channels expressed in HEK293 cells.J Physiol. 2006 Jan 15;570(Pt 2):219-35. doi: 10.1113/jphysiol.2005.097998. Epub 2005 Nov 10. J Physiol. 2006. PMID: 16284075 Free PMC article.
-
TRPC5.Handb Exp Pharmacol. 2014;222:129-56. doi: 10.1007/978-3-642-54215-2_6. Handb Exp Pharmacol. 2014. PMID: 24756705 Review.
-
Mechanosensory ion channels in charophyte cells: the response to touch and salinity stress.Eur Biophys J. 2002 Sep;31(5):341-55. doi: 10.1007/s00249-002-0222-6. Epub 2002 Jun 13. Eur Biophys J. 2002. PMID: 12202910 Review.
Cited by
-
Inhibition of Canonical Transient Receptor Potential Channels 4/5 with Highly Selective and Potent Small-Molecule HC-070 Alleviates Mechanical Hypersensitivity in Rat Models of Visceral and Neuropathic Pain.Int J Mol Sci. 2023 Feb 8;24(4):3350. doi: 10.3390/ijms24043350. Int J Mol Sci. 2023. PMID: 36834762 Free PMC article.
-
Cardiac vagal afferent neurotransmission in health and disease: review and knowledge gaps.Front Neurosci. 2023 Jun 7;17:1192188. doi: 10.3389/fnins.2023.1192188. eCollection 2023. Front Neurosci. 2023. PMID: 37351426 Free PMC article. Review.
-
Tension at the gate: sensing mechanical forces at the blood-brain barrier in health and disease.J Neuroinflammation. 2024 Dec 18;21(1):325. doi: 10.1186/s12974-024-03321-2. J Neuroinflammation. 2024. PMID: 39696463 Free PMC article. Review.
-
Mechanosensitive ion channels in cell migration.Cells Dev. 2021 Jun;166:203683. doi: 10.1016/j.cdev.2021.203683. Epub 2021 Apr 27. Cells Dev. 2021. PMID: 33994356 Free PMC article. Review.
-
TRPC5 deletion in the central amygdala antagonizes high-fat diet-induced obesity by increasing sympathetic innervation.Int J Obes (Lond). 2022 Aug;46(8):1544-1555. doi: 10.1038/s41366-022-01151-x. Epub 2022 May 20. Int J Obes (Lond). 2022. PMID: 35589963
References
-
- Cantor RS. The lateral pressure profile in membranes: a physical mechanism of general anesthesia. Biochemistry. 1997;36(9):2339–44. Epub 1997/03/04. 10.1021/bi9627323 bi9627323 [pii]. . - PubMed
-
- Hamill OP, Martinac B. Molecular basis of mechanotransduction in living cells. Physiol Rev. 2001;81(2):685–740. Epub 2001/03/29. . - PubMed
-
- Kung C. A possible unifying principle for mechanosensation. Nature. 2005;436(7051):647–54. Epub 2005/08/05. nature03896 [pii] 10.1038/nature03896. . - PubMed
-
- Pedersen SF, Nilius B. Transient receptor potential channels in mechanosensing and cell volume regulation. Methods Enzymol. 2007;428:183–207. Epub 2007/09/19. S0076-6879(07)28010-3 [pii] 10.1016/S0076-6879(07)28010-3. . - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

