Inhibition of BET bromodomains as a therapeutic strategy for cancer drug discovery
- PMID: 25849938
- PMCID: PMC4467383
- DOI: 10.18632/oncotarget.3551
Inhibition of BET bromodomains as a therapeutic strategy for cancer drug discovery
Abstract
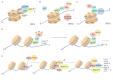
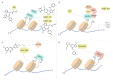
As a conserved protein interaction module that recognizes and binds to acetylated lysine, bromodomain (BRD) contains a deep, largely hydrophobic acetyl lysine binding site. Proteins that share the feature of containing two BRDs and an extra-terminal domain belong to BET family, including BRD2, BRD3, BRD4 and BRDT. BET family proteins perform transcription regulatory function under normal conditions, while in cancer, they regulate transcription of several oncogenes, such as c-Myc and Bcl-2. Thus, targeting BET proteins may be a promising strategy, and intense interest of BET proteins has fueled the development of structure-based bromodomain inhibitors in cancer. In this review, we focus on summarizing several small-molecule BET inhibitors and their relevant anti-tumor mechanisms, which would provide a clue for exploiting new targeted BET inhibitors in the future cancer therapy.
Keywords: BET inhibitor; BRD2/4; BRD3; BRDT; bromodomain.
Conflict of interest statement
None.
Figures


References
-
- Choudhary C, Kumar C, Gnad F, Nielsen ML, Rehman M, Walther TC, Olsen JV, Mann M. Lysine acetylationtargets protein complexes and co-regulates major cellular functions. Science. 2009;325:834–840. - PubMed
-
- Kuo MH, Allis CD. Roles of histone acetyltransferases and deacetylases in gene regulation. Bioessays. 1998;20:615–626. - PubMed
-
- Filippakopoulos P, Knapp S. Targeting bromodomains: epigenetic readers of lysine acetylation. Nat Rev Drug Discov. 2014;13:337–356. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

