PKCε and allopregnanolone: functional cross-talk at the GABAA receptor level
- PMID: 25852476
- PMCID: PMC4365694
- DOI: 10.3389/fncel.2015.00083
PKCε and allopregnanolone: functional cross-talk at the GABAA receptor level
Abstract
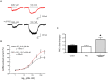
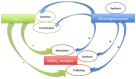
Changes in GABAergic inhibition occur during physiological processes, during response to drugs and in various pathologies. These changes can be achieved through direct allosteric modifications at the γ-amino butyric acid (GABA) type A (GABAA) receptor protein level, or by altering the synthesis, trafficking and stability of the receptor. Neurosteroids (NSs) and protein kinase C (PKC) are potent modulators of GABAA receptors and their effects are presumably intermingled, even though evidence for this hypothesis is only partially explored. However, several PKC isoforms are able to phosphorylate the GABAA receptor, producing different functional effects. We focused on the ε isoform, that has been correlated to the sensitivity of the GABAA receptor to allosteric modulators and whose expression may be regulated in peripheral sensory neurons by NSs. The cross-talk between PKC-ε and NSs, leading to changes in GABAA receptor functionality, is considered and discussed in this perspective.
Keywords: GABAA receptor; PKCepsilon; neurosteroids; phosphorylation site; receptor traffiking.
Figures
Similar articles
-
Modulation of GABAA receptor-mediated IPSCs by neuroactive steroids in a rat hypothalamo-hypophyseal coculture model.J Physiol. 1997 Apr 15;500 ( Pt 2)(Pt 2):475-85. doi: 10.1113/jphysiol.1997.sp022034. J Physiol. 1997. PMID: 9147331 Free PMC article.
-
Cross-talk between allosteric and orthosteric binding sites of γ-amino butyric acid type A receptors (GABAA-Rs): A computational study revealing the structural basis of selectivity.J Biomol Struct Dyn. 2019 Aug;37(12):3065-3080. doi: 10.1080/07391102.2018.1508367. Epub 2019 Jan 4. J Biomol Struct Dyn. 2019. PMID: 30608219
-
Developmentally regulated neurosteroid synthesis enhances GABAergic neurotransmission in mouse thalamocortical neurones.J Physiol. 2015 Jan 1;593(1):267-84. doi: 10.1113/jphysiol.2014.280263. Epub 2014 Dec 3. J Physiol. 2015. PMID: 25556800 Free PMC article.
-
Short-term modulation of GABAA receptor function in the adult female rat.Prog Brain Res. 2002;139:31-42. doi: 10.1016/s0079-6123(02)39005-8. Prog Brain Res. 2002. PMID: 12436924 Review.
-
From GABAA receptor diversity emerges a unified vision of GABAergic inhibition.Annu Rev Pharmacol Toxicol. 1998;38:321-50. doi: 10.1146/annurev.pharmtox.38.1.321. Annu Rev Pharmacol Toxicol. 1998. PMID: 9597158 Review.
Cited by
-
Schwann cell development, maturation and regeneration: a focus on classic and emerging intracellular signaling pathways.Neural Regen Res. 2017 Jul;12(7):1013-1023. doi: 10.4103/1673-5374.211172. Neural Regen Res. 2017. PMID: 28852375 Free PMC article. Review.
-
Editorial on "New perspectives in neurosteroids action: a special player allopregnanolone".Front Cell Neurosci. 2015 Apr 9;9:133. doi: 10.3389/fncel.2015.00133. eCollection 2015. Front Cell Neurosci. 2015. PMID: 25914624 Free PMC article. No abstract available.
-
GABA-ergic Modulators: New Therapeutic Approaches to Premenstrual Dysphoric Disorder.CNS Drugs. 2023 Aug;37(8):679-693. doi: 10.1007/s40263-023-01030-7. Epub 2023 Aug 5. CNS Drugs. 2023. PMID: 37542704 Review.
-
Role of GABAAR in the Transition From Acute to Chronic Pain and the Analgesic Effect of Electroacupuncture on Hyperalgesic Priming Model Rats.Front Neurosci. 2021 Jun 17;15:691455. doi: 10.3389/fnins.2021.691455. eCollection 2021. Front Neurosci. 2021. PMID: 34220444 Free PMC article.
-
Physiopathological Role of Neuroactive Steroids in the Peripheral Nervous System.Int J Mol Sci. 2020 Nov 26;21(23):9000. doi: 10.3390/ijms21239000. Int J Mol Sci. 2020. PMID: 33256238 Free PMC article. Review.
References
-
- Alipour M. R., Aliparasti M. R., Keyhanmanesh R., Almasi S., Halimi M., Ansarin K., et al. . (2011). Effect of ghrelin on protein kinase C-ε and protein kinase C-δ gene expression in the pulmonary arterial smooth muscles of chronic hypoxic rats. J. Endocrinol. Invest. 34, e369–e373. 10.3275/8056 - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

