Imbalance of the nerve growth factor and its precursor as a potential biomarker for diabetic retinopathy
- PMID: 25853140
- PMCID: PMC4380101
- DOI: 10.1155/2015/571456
Imbalance of the nerve growth factor and its precursor as a potential biomarker for diabetic retinopathy
Abstract
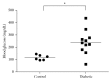
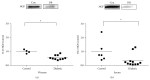
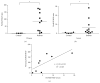
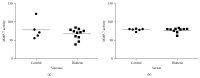
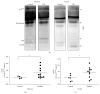
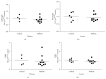
Our previous studies have demonstrated that diabetes-induced oxidative stress alters homeostasis of retinal nerve growth factor (NGF) resulting in accumulation of its precursor, proNGF, at the expense of NGF which plays a critical role in preserving neuronal and retinal function. This imbalance coincided with retinal damage in experimental diabetes. Here we test the hypothesis that alteration of proNGF and NGF levels observed in retina and vitreous will be mirrored in serum of diabetic patients. Blood and vitreous samples were collected from patients (diabetic and nondiabetic) undergoing vitrectomy at Georgia Regents University under approved IRB. Levels of proNGF, NGF, and p75(NTR) shedding were detected using Western blot analysis. MMP-7 activity was also assayed. Diabetes-induced proNGF expression and impaired NGF expression were observed in vitreous and serum. Vitreous and sera from diabetic patients (n = 11) showed significant 40.8-fold and 3.6-fold increases, respectively, compared to nondiabetics (n = 9). In contrast, vitreous and sera from diabetic patients showed significant 44% and 64% reductions in NGF levels, respectively, compared to nondiabetics. ProNGF to NGF ratios showed significant correlation between vitreous and serum. Further characterization of diabetes-induced imbalance in the proNGF to NGF ratio will facilitate its utility as an early biomarker for diabetic complications.
Figures

Similar articles
-
Modulation of p75(NTR) prevents diabetes- and proNGF-induced retinal inflammation and blood-retina barrier breakdown in mice and rats.Diabetologia. 2013 Oct;56(10):2329-39. doi: 10.1007/s00125-013-2998-6. Epub 2013 Aug 7. Diabetologia. 2013. PMID: 23918145 Free PMC article.
-
Modulation of the p75 neurotrophin receptor using LM11A-31 prevents diabetes-induced retinal vascular permeability in mice via inhibition of inflammation and the RhoA kinase pathway.Diabetologia. 2019 Aug;62(8):1488-1500. doi: 10.1007/s00125-019-4885-2. Epub 2019 May 9. Diabetologia. 2019. PMID: 31073629 Free PMC article.
-
Pronerve growth factor induces angiogenesis via activation of TrkA: possible role in proliferative diabetic retinopathy.J Diabetes Res. 2013;2013:432659. doi: 10.1155/2013/432659. Epub 2013 Aug 12. J Diabetes Res. 2013. PMID: 23998130 Free PMC article.
-
Expression and signaling of NGF in the healthy and injured retina.Cytokine Growth Factor Rev. 2017 Apr;34:43-57. doi: 10.1016/j.cytogfr.2016.11.005. Epub 2016 Dec 2. Cytokine Growth Factor Rev. 2017. PMID: 27964967 Review.
-
Nerve growth factor in metabolic complications and Alzheimer's disease: Physiology and therapeutic potential.Biochim Biophys Acta Mol Basis Dis. 2020 Oct 1;1866(10):165858. doi: 10.1016/j.bbadis.2020.165858. Epub 2020 Jun 10. Biochim Biophys Acta Mol Basis Dis. 2020. PMID: 32531260 Review.
Cited by
-
QiMing granules for diabetic retinopathy: a systematic review and meta-analysis of randomized controlled trials.Front Pharmacol. 2024 Aug 22;15:1429071. doi: 10.3389/fphar.2024.1429071. eCollection 2024. Front Pharmacol. 2024. PMID: 39239647 Free PMC article.
-
Deletion of p75NTR prevents vaso-obliteration and retinal neovascularization via activation of Trk- A receptor in ischemic retinopathy model.Sci Rep. 2018 Aug 21;8(1):12490. doi: 10.1038/s41598-018-30029-0. Sci Rep. 2018. PMID: 30131506 Free PMC article.
-
Effect of miR-200b on retinal endothelial cell function under high glucose environment.Int J Clin Exp Pathol. 2015 Sep 1;8(9):10482-7. eCollection 2015. Int J Clin Exp Pathol. 2015. PMID: 26617758 Free PMC article.
-
Carbamazepine Alleviates Retinal and Optic Nerve Neural Degeneration in Diabetic Mice via Nerve Growth Factor-Induced PI3K/Akt/mTOR Activation.Front Neurosci. 2019 Nov 1;13:1089. doi: 10.3389/fnins.2019.01089. eCollection 2019. Front Neurosci. 2019. PMID: 31736682 Free PMC article.
-
Deletion of the Neurotrophin Receptor p75NTR Prevents Diabetes-Induced Retinal Acellular Capillaries in Streptozotocin-Induced Mouse Diabetic Model.J Diabetes Metab Disord Control. 2017;4(6):129. doi: 10.15406/jdmdc.2017.04.00129. Epub 2017 Dec 21. J Diabetes Metab Disord Control. 2017. PMID: 29658956 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

