Fibrin gel as an injectable biodegradable scaffold and cell carrier for tissue engineering
- PMID: 25853146
- PMCID: PMC4380102
- DOI: 10.1155/2015/685690
Fibrin gel as an injectable biodegradable scaffold and cell carrier for tissue engineering
Abstract
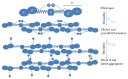
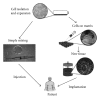
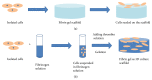
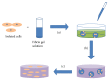
Due to the increasing needs for organ transplantation and a universal shortage of donated tissues, tissue engineering emerges as a useful approach to engineer functional tissues. Although different synthetic materials have been used to fabricate tissue engineering scaffolds, they have many limitations such as the biocompatibility concerns, the inability to support cell attachment, and undesirable degradation rate. Fibrin gel, a biopolymeric material, provides numerous advantages over synthetic materials in functioning as a tissue engineering scaffold and a cell carrier. Fibrin gel exhibits excellent biocompatibility, promotes cell attachment, and can degrade in a controllable manner. Additionally, fibrin gel mimics the natural blood-clotting process and self-assembles into a polymer network. The ability for fibrin to cure in situ has been exploited to develop injectable scaffolds for the repair of damaged cardiac and cartilage tissues. Additionally, fibrin gel has been utilized as a cell carrier to protect cells from the forces during the application and cell delivery processes while enhancing the cell viability and tissue regeneration. Here, we review the recent advancement in developing fibrin-based biomaterials for the development of injectable tissue engineering scaffold and cell carriers.
Figures



Similar articles
-
Computational Fluid Dynamics Modeling of Material Transport Through Triply Periodic Minimal Surface Scaffolds for Bone Tissue Engineering.J Biomech Eng. 2025 Mar 1;147(3):031007. doi: 10.1115/1.4067575. J Biomech Eng. 2025. PMID: 39790065
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Evaluation of biological protein-based collagen scaffolds in cartilage and musculoskeletal tissue engineering--a systematic review of the literature.Curr Stem Cell Res Ther. 2012 Jul;7(4):302-9. doi: 10.2174/157488812800793045. Curr Stem Cell Res Ther. 2012. PMID: 22563667
-
Keratin-containing scaffolds for tissue engineering applications: a review.J Biomater Sci Polym Ed. 2024 Apr;35(6):916-965. doi: 10.1080/09205063.2024.2311450. Epub 2024 Feb 13. J Biomater Sci Polym Ed. 2024. PMID: 38349200 Review.
-
Vat photo-polymerization 3D printing of gradient scaffolds for osteochondral tissue regeneration.Acta Biomater. 2025 Jun 15;200:67-86. doi: 10.1016/j.actbio.2025.05.042. Epub 2025 May 23. Acta Biomater. 2025. PMID: 40414264 Review.
Cited by
-
Endostatin in 3D Fibrin Hydrogel Scaffolds Promotes Chondrogenic Differentiation in Swine Neonatal Meniscal Cells.Biomedicines. 2022 Sep 27;10(10):2415. doi: 10.3390/biomedicines10102415. Biomedicines. 2022. PMID: 36289678 Free PMC article.
-
Hydrogels as artificial matrices for cell seeding in microfluidic devices.RSC Adv. 2020 Dec 8;10(71):43682-43703. doi: 10.1039/d0ra08566a. eCollection 2020 Nov 27. RSC Adv. 2020. PMID: 35519701 Free PMC article. Review.
-
Composite bijel-templated hydrogels for cell delivery.ACS Biomater Sci Eng. 2018 Feb 12;4(2):587-594. doi: 10.1021/acsbiomaterials.7b00809. Epub 2018 Jan 18. ACS Biomater Sci Eng. 2018. PMID: 30555892 Free PMC article.
-
Screening of biopolymeric materials for cardiovascular surgery toxicity-Evaluation of their surface relief with assessment of morphological aspects of monocyte/macrophage polarization in atherosclerosis patients.Toxicol Rep. 2018 Nov 16;6:74-90. doi: 10.1016/j.toxrep.2018.11.009. eCollection 2019. Toxicol Rep. 2018. PMID: 30581762 Free PMC article.
-
Delivery of silver sulfadiazine and adipose derived stem cells using fibrin hydrogel improves infected burn wound regeneration.PLoS One. 2019 Jun 13;14(6):e0217965. doi: 10.1371/journal.pone.0217965. eCollection 2019. PLoS One. 2019. PMID: 31194776 Free PMC article.
References
-
- U. S. Department of Health & Human Services. http://www.organdonor.gov/ - PubMed
-
- Gunatillake P. A., Adhikari R. Biodegradable synthetic polymers for tissue engineering. European Cells and Materials. 2003;5:1–16. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

