Cell surface-based sensing with metallic nanoparticles
- PMID: 25853985
- PMCID: PMC4478158
- DOI: 10.1039/c4cs00387j
Cell surface-based sensing with metallic nanoparticles
Abstract
Metallic nanoparticles provide versatile scaffolds for biosensing applications. In this review, we focus on the use of metallic nanoparticles for cell surface sensings. Examples of the use of both specific recognition and array-based "chemical nose" approaches to cell surface sensing will be discussed.
Figures
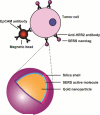
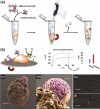
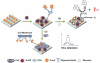





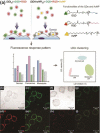

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

