LP99: Discovery and Synthesis of the First Selective BRD7/9 Bromodomain Inhibitor
- PMID: 25864491
- PMCID: PMC4449114
- DOI: 10.1002/anie.201501394
LP99: Discovery and Synthesis of the First Selective BRD7/9 Bromodomain Inhibitor
Abstract
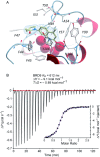
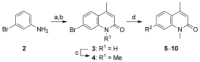
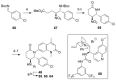
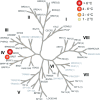
The bromodomain-containing proteins BRD9 and BRD7 are part of the human SWI/SNF chromatin-remodeling complexes BAF and PBAF. To date, no selective inhibitor for BRD7/9 has been reported despite its potential value as a biological tool or as a lead for future therapeutics. The quinolone-fused lactam LP99 is now reported as the first potent and selective inhibitor of the BRD7 and BRD9 bromodomains. Development of LP99 from a fragment hit was expedited through balancing structure-based inhibitor design and biophysical characterization against tractable chemical synthesis: Complexity-building nitro-Mannich/lactamization cascade processes allowed for early structure-activity relationship studies whereas an enantioselective organocatalytic nitro-Mannich reaction enabled the synthesis of the lead scaffold in enantioenriched form and on scale. This epigenetic probe was shown to inhibit the association of BRD7 and BRD9 to acetylated histones in vitro and in cells. Moreover, LP99 was used to demonstrate that BRD7/9 plays a role in regulating pro-inflammatory cytokine secretion.
Keywords: bromodomain; cascade reactions; enantioselective catalysis; epigenetics; organocatalysis.
© 2015 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA. This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Figures



References
-
- Wu WJ, Hu KS, Chen DL, Zeng ZL, Luo HY, Wang F, Wang DS, Wang ZQ, He F, Xu RH. Eur. J. Clin. Invest. 2013;43:131–140. - PubMed
-
- Drost J, et al. Nat. Cell Biol. 2010;12:380–389. - PubMed
-
- Harte MT, O′Brien GJ, Ryan NM, Gorski JJ, Savage KI, Crawford NT, Mullan PB, Harkin DP. Cancer Res. 2010;70:2538–2547. - PubMed
-
- B. Zhu et al., Mol. Carcinog2014DOI: doi/10.1002/mc.22140.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Chemical Information
Molecular Biology Databases

