Biochemical Validation of the Glyoxylate Cycle in the Cyanobacterium Chlorogloeopsis fritschii Strain PCC 9212
- PMID: 25869135
- PMCID: PMC4447974
- DOI: 10.1074/jbc.M115.648170
Biochemical Validation of the Glyoxylate Cycle in the Cyanobacterium Chlorogloeopsis fritschii Strain PCC 9212
Abstract
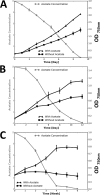
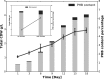
Cyanobacteria are important photoautotrophic bacteria with extensive but variable metabolic capacities. The existence of the glyoxylate cycle, a variant of the TCA cycle, is still poorly documented in cyanobacteria. Previous studies reported the activities of isocitrate lyase and malate synthase, the key enzymes of the glyoxylate cycle in some cyanobacteria, but other studies concluded that these enzymes are missing. In this study the genes encoding isocitrate lyase and malate synthase from Chlorogloeopsis fritschii PCC 9212 were identified, and the recombinant enzymes were biochemically characterized. Consistent with the presence of the enzymes of the glyoxylate cycle, C. fritschii could assimilate acetate under both light and dark growth conditions. Transcript abundances for isocitrate lyase and malate synthase increased, and C. fritschii grew faster, when the growth medium was supplemented with acetate. Adding acetate to the growth medium also increased the yield of poly-3-hydroxybutyrate. When the genes encoding isocitrate lyase and malate synthase were expressed in Synechococcus sp. PCC 7002, the acetate assimilation capacity of the resulting strain was greater than that of wild type. Database searches showed that the genes for the glyoxylate cycle exist in only a few other cyanobacteria, all of which are able to fix nitrogen. This study demonstrates that the glyoxylate cycle exists in a few cyanobacteria, and that this pathway plays an important role in the assimilation of acetate for growth in one of those organisms. The glyoxylate cycle might play a role in coordinating carbon and nitrogen metabolism under conditions of nitrogen fixation.
Keywords: TCA cycle; acetyl coenzyme A (acetyl-CoA); cyanobacteria; glyoxylate cycle; isocitrate lyase; malate synthase; metabolism; photosynthesis; poly-3-hydroxybutyrate; tricarboxylic acid cycle (TCA cycle) (Krebs cycle).
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures





References
-
- Pinchuk G. E., Geydebrekht O. V., Hill E. A., Reed J. L., Konopka A. E., Beliaev A. S., Fredrickson J. K. (2011) Pyruvate and lactate metabolism by Shewanella oneidensis MR-1 under fermentation, oxygen limitation, and fumarate respiration conditions. Appl. Environ. Microbiol. 77, 8234–8240 - PMC - PubMed
-
- Brown T. D., Jones-Mortimer M. C., Kornberg H. L. (1977) The enzymic interconversion of acetate and acetyl-coenzyme A in Escherichia coli. J. Gen. Microbiol. 102, 327–336 - PubMed
-
- Ihlenfeldt M. J., Gibson J. (1977) Acetate uptake by the unicellular cyanobacteria Synechococcus and Aphanocapsa. Arch. Microbiol. 113, 231–241 - PubMed
-
- Kay W. W., Kornberg H. L. (1971) The uptake of C4-dicarboxylic acids by Escherichia coli. Eur. J. Biochem. 18, 274–281 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

