The GLP-1 Receptor Agonist Liraglutide Improves Memory Function and Increases Hippocampal CA1 Neuronal Numbers in a Senescence-Accelerated Mouse Model of Alzheimer's Disease
- PMID: 25869785
- PMCID: PMC4878312
- DOI: 10.3233/JAD-143090
The GLP-1 Receptor Agonist Liraglutide Improves Memory Function and Increases Hippocampal CA1 Neuronal Numbers in a Senescence-Accelerated Mouse Model of Alzheimer's Disease
Abstract
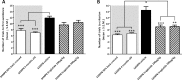
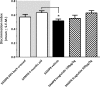
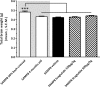
Recent studies indicate that glucagon-like peptide 1 (GLP-1) receptor agonists, currently used in the management of type 2 diabetes, exhibit neurotrophic and neuroprotective effects in amyloid-β (Aβ) toxicity models of Alzheimer's disease (AD). We investigated the potential pro-cognitive and neuroprotective effects of the once-daily GLP-1 receptor agonist liraglutide in senescence-accelerated mouse prone 8 (SAMP8) mice, a model of age-related sporadic AD not dominated by amyloid plaques. Six-month-old SAMP8 mice received liraglutide (100 or 500 μg/kg/day, s.c.) or vehicle once daily for 4 months. Vehicle-dosed age-matched 50% back-crossed as well as untreated young (4-month-old) SAMP8 mice were used as control groups for normal memory function. Vehicle-dosed 10-month-old SAMP8 mice showed significant learning and memory retention deficits in an active-avoidance T-maze, as compared to both control groups. Also, 10-month-old SAMP8 mice displayed no immunohistological signatures of amyloid-β plaques or hyperphosphorylated tau, indicating the onset of cognitive deficits prior to deposition of amyloid plaques and neurofibrillary tangles in this AD model. Liraglutide significantly increased memory retention and total hippocampal CA1 pyramidal neuron numbers in SAMP8 mice, as compared to age-matched vehicle-dosed SAMP8 mice. In conclusion, liraglutide delayed or partially halted the progressive decline in memory function associated with hippocampal neuronal loss in a mouse model of pathological aging with characteristics of neurobehavioral and neuropathological impairments observed in early-stage sporadic AD.
Keywords: Alzheimer’s disease; GLP-1 receptor agonist; SAMP8 mouse; hippocampus; liraglutide; memory function; neuroprotection; stereology.
Figures


Similar articles
-
Effects of accelerated senescence on learning and memory, locomotion and anxiety-like behavior in APP/PS1 mouse model of Alzheimer's disease.J Neurol Sci. 2013 Dec 15;335(1-2):145-54. doi: 10.1016/j.jns.2013.09.018. Epub 2013 Sep 21. J Neurol Sci. 2013. PMID: 24095271
-
Subcutaneous administration of liraglutide ameliorates learning and memory impairment by modulating tau hyperphosphorylation via the glycogen synthase kinase-3β pathway in an amyloid β protein induced alzheimer disease mouse model.Eur J Pharmacol. 2016 Jul 15;783:23-32. doi: 10.1016/j.ejphar.2016.04.052. Epub 2016 Apr 27. Eur J Pharmacol. 2016. PMID: 27131827
-
The behavioral, pathological and therapeutic features of the senescence-accelerated mouse prone 8 strain as an Alzheimer's disease animal model.Ageing Res Rev. 2014 Jan;13:13-37. doi: 10.1016/j.arr.2013.10.002. Epub 2013 Nov 21. Ageing Res Rev. 2014. PMID: 24269312
-
SAMP8 Mice as a Model of Age-Related Cognition Decline with Underlying Mechanisms in Alzheimer's Disease.J Alzheimers Dis. 2020;75(2):385-395. doi: 10.3233/JAD-200063. J Alzheimers Dis. 2020. PMID: 32310176 Review.
-
Senescence accelerated mouse-prone 8: a model of neuroinflammation and aging with features of sporadic Alzheimer's disease.Stem Cells. 2025 Feb 12;43(2):sxae091. doi: 10.1093/stmcls/sxae091. Stem Cells. 2025. PMID: 39813151 Free PMC article. Review.
Cited by
-
Diabetes Mellitus: A Path to Amnesia, Personality, and Behavior Change.Biology (Basel). 2022 Feb 28;11(3):382. doi: 10.3390/biology11030382. Biology (Basel). 2022. PMID: 35336756 Free PMC article. Review.
-
Liraglutide Protects Against Brain Amyloid-β1-42 Accumulation in Female Mice with Early Alzheimer's Disease-Like Pathology by Partially Rescuing Oxidative/Nitrosative Stress and Inflammation.Int J Mol Sci. 2020 Mar 4;21(5):1746. doi: 10.3390/ijms21051746. Int J Mol Sci. 2020. PMID: 32143329 Free PMC article.
-
A review of type 2 diabetes mellitus and cognitive impairment.Front Endocrinol (Lausanne). 2025 Aug 4;16:1624472. doi: 10.3389/fendo.2025.1624472. eCollection 2025. Front Endocrinol (Lausanne). 2025. PMID: 40831951 Free PMC article. Review.
-
Artemisinin ameliorates cognitive decline by inhibiting hippocampal neuronal ferroptosis via Nrf2 activation in T2DM mice.Mol Med. 2024 Mar 7;30(1):35. doi: 10.1186/s10020-024-00797-9. Mol Med. 2024. PMID: 38454322 Free PMC article.
-
Interactions between metabolic, reward and cognitive processes in appetite control: Implications for novel weight management therapies.J Psychopharmacol. 2017 Nov;31(11):1460-1474. doi: 10.1177/0269881117736917. Epub 2017 Oct 26. J Psychopharmacol. 2017. PMID: 29072515 Free PMC article. Review.
References
-
- Querfurth HW, LaFerla FM. Alzheimer’s disease. N Engl J Med. 2010;362:329–344. - PubMed
-
- Rönnemaa E, Zethelius B, Sundelöf J, Sundström J, Degerman-Gunnarsson M, Berne C, Lannfelt L, Kilander L. Impaired insulin secretion increases the risk of Alzheimer disease. Neurology. 2008;71:1065–1071. - PubMed
-
- Moloney AM, Griffin RJ, Timmons S, O’Connor R, Ravid R, O’Neill C. Defects in IGF-1 receptor, insulin receptor and IRS-1/2 in Alzheimer’s disease indicate possible resistance to IGF-1 and insulin signalling. Neurobiol Aging. 2010;31:224–243. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

