ROS produced by NOX2 control in vitro development of cerebellar granule neurons development
- PMID: 25873309
- PMCID: PMC4720178
- DOI: 10.1177/1759091415578712
ROS produced by NOX2 control in vitro development of cerebellar granule neurons development
Abstract
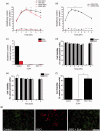
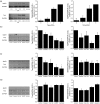
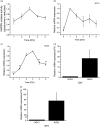
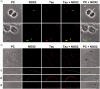
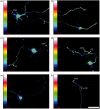
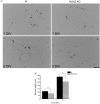
Reactive oxygen species (ROS) act as signaling molecules that regulate nervous system physiology. ROS have been related to neural differentiation, neuritogenesis, and programmed cell death. Nevertheless, little is known about the mechanisms involved in the regulation of ROS during neuronal development. In this study, we evaluated the mechanisms by which ROS are regulated during neuronal development and the implications of these molecules in this process. Primary cultures of cerebellar granule neurons (CGN) were used to address these issues. Our results show that during the first 3 days of CGN development in vitro (days in vitro; DIV), the levels of ROS increased, reaching a peak at 2 and 3 DIV under depolarizing (25 mM KCl) and nondepolarizing (5 mM KCl) conditions. Subsequently, under depolarizing conditions, the ROS levels markedly decreased, but in nondepolarizing conditions, the ROS levels increased gradually. This correlated with the extent of CGN maturation. Also, antioxidants and NADPH-oxidases (NOX) inhibitors reduced the expression of Tau and MAP2. On the other hand, the levels of glutathione markedly increased at 1 DIV. We inferred that the ROS increase at this time is critical for cell survival because glutathione depletion leads to axonal degeneration and CGN death only at 2 DIV. During the first 3 DIV, NOX2 was upregulated and expressed in filopodia and growth cones, which correlated with the hydrogen peroxide (H2O2) distribution in the cell. Finally, NOX2 KO CGN showed shorter neurites than wild-type CGN. Taken together, these results suggest that the regulation of ROS is critical during the early stages of CGN development.
Keywords: NADPH-oxidases; axonal morphogenesis; cerebellar granule neurons; glutathione; neuronal development; reactive oxygen species.
© The Author(s) 2015.
Figures






References
-
- Avshalumov M. V., Chen B. T., Koos T., Tepper J. M., Rice M. E. (2005) Endogenous hydrogen peroxide regulates the excitability of midbrain dopamine neurons via ATP-sensitive potassium channels. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience 25: 4222–4231. - PMC - PubMed
-
- Baker K., Marcus C. B., Huffman K., Kruk H., Malfroy B., Doctrow S. R. (1998) Synthetic combined superoxide dismutase/catalase mimetics are protective as a delayed treatment in a rat stroke model: A key role for reactive oxygen species in ischemic brain injury. The Journal of Pharmacology and Experimental Therapeutics 284: 215–221. - PubMed
-
- Barnham K. J., Masters C. L., Bush A. I. (2004) Neurodegenerative diseases and oxidative stress. Nature Reviews. Drug Discovery 3: 205–214. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous
