Inhibition of inflammatory arthritis using fullerene nanomaterials
- PMID: 25879437
- PMCID: PMC4400016
- DOI: 10.1371/journal.pone.0126290
Inhibition of inflammatory arthritis using fullerene nanomaterials
Abstract
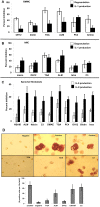
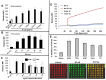
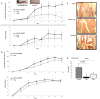
Inflammatory arthritis (e.g. rheumatoid arthritis; RA) is a complex disease driven by the interplay of multiple cellular lineages. Fullerene derivatives have previously been shown to have anti-inflammatory capabilities mediated, in part, by their ability to prevent inflammatory mediator release by mast cells (MC). Recognizing that MC can serve as a cellular link between autoantibodies, soluble mediators, and other effector populations in inflammatory arthritis, it was hypothesized that fullerene derivatives might be used to target this inflammatory disease. A panel of fullerene derivatives was tested for their ability to affect the function of human skin-derived MC as well as other lineages implicated in arthritis, synovial fibroblasts and osteoclasts. It is shown that certain fullerene derivatives blocked FcγR- and TNF-α-induced mediator release from MC; TNF-α-induced mediator release from RA synovial fibroblasts; and maturation of human osteoclasts. MC inhibition by fullerene derivatives was mediated through the reduction of mitochondrial membrane potential and FcγR-mediated increases in cellular reactive oxygen species and NF-κB activation. Based on these in vitro data, two fullerene derivatives (ALM and TGA) were selected for in vivo studies using K/BxN serum transfer arthritis in C57BL/6 mice and collagen-induced arthritis (CIA) in DBA/1 mice. Dye-conjugated fullerenes confirmed localization to affected joints in arthritic animals but not in healthy controls. In the K/BxN moldel, fullerenes attenuated arthritis, an effect accompanied by reduced histologic inflammation, cartilage/bone erosion, and serum levels of TNF-α. Fullerenes remained capable of attenuating K/BxN arthritis in mast cell-deficient mice Cre-Master mice, suggesting that lineages beyond the MC represent relevant targets in this system. These studies suggest that fullerene derivatives may hold promise both as an assessment tool and as anti-inflammatory therapy of arthritis.
Conflict of interest statement
Figures

Comment in
-
Experimental arthritis: Fullerene nanoparticles ameliorate disease in arthritis mouse model.Nat Rev Rheumatol. 2015 Jun;11(6):319. doi: 10.1038/nrrheum.2015.62. Epub 2015 May 5. Nat Rev Rheumatol. 2015. PMID: 25939420 No abstract available.
References
-
- Davies KJ. Oxidative stress: the paradox of aerobic life. BiochemSocSymp. 1995;61: 1–31. - PubMed
-
- Park H, Bourla AB, Kastner DL, Colbert RA, Siegel RM. Lighting the fires within: the cell biology of autoinflammatory diseases. Nat Rev Immunol. 2012;12: 570–580. doi: 510.1038/nri3261 - PMC - PubMed
-
- Arab HH, El-Sawalhi MM. Carvedilol alleviates adjuvant-induced arthritis and subcutaneous air pouch edema: Modulation of oxidative stress and inflammatory mediators. Toxicol Appl Pharmacol. 2013;27: 00040–00049. - PubMed
-
- Drafi F, Bauerova K, Kuncirova V, Ponist S, Mihalova D, Fedorova T, et al. Pharmacological influence on processes of adjuvant arthritis: Effect of the combination of an antioxidant active substance with methotrexate. Interdiscip Toxicol. 2012;5: 84–91. doi: 10.2478/v10102-10012-10015-10104 - DOI - PMC - PubMed
-
- Zhang W, Dai SM. Mechanisms involved in the therapeutic effects of Paeonia lactiflora Pallas in rheumatoid arthritis. Int Immunopharmacol. 2012;14: 27–31. doi: 10.1016/j.intimp.2012.1006.1001 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

