Use of Saliva to Identify Varicella Zoster Virus Infection of the Gut
- PMID: 25882301
- PMCID: PMC4607733
- DOI: 10.1093/cid/civ320
Use of Saliva to Identify Varicella Zoster Virus Infection of the Gut
Abstract
Background: Varicella zoster virus (VZV) establishes latency in dorsal root, cranial nerve, and enteric ganglia and can reactivate to cause zoster. Serious gastrointestinal dysfunction can result from VZV reactivation in enteric neurons (enteric zoster), but an absence of rash makes diagnosis difficult. We thus determined whether detecting VZV DNA in saliva facilitates identification of enteric zoster.
Methods: Nested and real-time polymerase chain reaction were used to validate salivary VZV DNA as a surrogate marker of VZV reactivation and then to determine the utility of that marker for the identification of those individuals within a population defined by abdominal pain that might have enteric zoster.
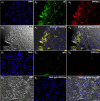
Results: Salivary VZV DNA was detected in 0 of 20 healthy negative controls, 11 of 16 positive controls with zoster or varicella (P < .0001), 2 of 2 patients with zoster sine herpete (P < .01), 6 of 11 patients with unexplained abdominal pain (P < .001), and 0 of 8 patients with unrelated gastrointestinal disorders. Salivary VZV DNA disappeared after recovery in 9 of 9 tested subjects with zoster, 2 of 2 with zoster sine herpete, and 5 of 5 with abdominal pain. One patient with abdominal pain and salivary VZV DNA had perforated gastric ulcers, necessitating a wedge gastrectomy. VZV DNA (vaccine type) was found in the resected stomach; immediate early (ORF63p) and late (gE) VZV proteins were immunocytochemically detected in gastric epithelium. After recovery, VZV DNA and proteins were not detected in gastric biopsies or saliva.
Conclusions: Detection of salivary VZV DNA in patients with abdominal pain helps to identify putative enteric zoster for investigation and treatment.
Keywords: abdominal pain; bleeding peptic ulcer; enteric infections; enteric nervous system; enteric neurons.
© The Author 2015. Published by Oxford University Press on behalf of the Infectious Diseases Society of America. All rights reserved. For Permissions, please e-mail: journals.permissions@oup.com.
Figures



References
-
- Gilden DH, Wright R, Schneck S, Gwaltney JM, Mahalingam R. Zoster sine herpete, a clinical variant. Ann Neurol 1994; 35:530–3. - PubMed
-
- Edelman DA, Antaki F, Basson MD, Salwen WA, Gruber SA, Losanoff JE. Ogilvie syndrome and herpes zoster: case report and review of the literature. J Emerg Med 2009; 39:696–700. - PubMed
-
- Pui JC, Furth EE, Minda J, Montone KT. Demonstration of varicella-zoster virus infection in the muscularis propria and myenteric plexi of the colon in an HIV-positive patient with herpes zoster and small bowel pseudo-obstruction (Ogilvie's syndrome). Am J Gastroenterol 2001; 96:1627–30. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

