Interactions between Plasmodium falciparum skeleton-binding protein 1 and the membrane skeleton of malaria-infected red blood cells
- PMID: 25883090
- PMCID: PMC4638388
- DOI: 10.1016/j.bbamem.2015.03.038
Interactions between Plasmodium falciparum skeleton-binding protein 1 and the membrane skeleton of malaria-infected red blood cells
Abstract
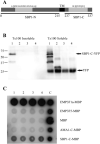
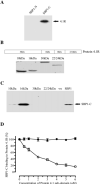
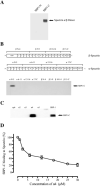
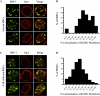
During development inside red blood cells (RBCs), Plasmodium falciparum malaria parasites export proteins that associate with the RBC membrane skeleton. These interactions cause profound changes to the biophysical properties of RBCs that underpin the often severe and fatal clinical manifestations of falciparum malaria. P. falciparum erythrocyte membrane protein 1 (PfEMP1) is one such exported parasite protein that plays a major role in malaria pathogenesis since its exposure on the parasitised RBC surface mediates their adhesion to vascular endothelium and placental syncytioblasts. En route to the RBC membrane skeleton, PfEMP1 transiently associates with Maurer's clefts (MCs), parasite-derived membranous structures in the RBC cytoplasm. We have previously shown that a resident MC protein, skeleton-binding protein 1 (SBP1), is essential for the placement of PfEMP1 onto the RBC surface and hypothesised that the function of SBP1 may be to target MCs to the RBC membrane. Since this would require additional protein interactions, we set out to identify binding partners for SBP1. Using a combination of approaches, we have defined the region of SBP1 that binds specifically to defined sub-domains of two major components of the RBC membrane skeleton, protein 4.1R and spectrin. We show that these interactions serve as one mechanism to anchor MCs to the RBC membrane skeleton, however, while they appear to be necessary, they are not sufficient for the translocation of PfEMP1 onto the RBC surface. The N-terminal domain of SBP1 that resides within the lumen of MCs clearly plays an essential, but presently unknown role in this process.
Keywords: Cell adhesion and mechanics; Malaria; Plasmodium falciparum; Protein 4.1R; Red blood cell; Spectrin.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures
Similar articles
-
A Maurer's cleft-associated protein is essential for expression of the major malaria virulence antigen on the surface of infected red blood cells.J Cell Biol. 2006 Mar 13;172(6):899-908. doi: 10.1083/jcb.200509122. Epub 2006 Mar 6. J Cell Biol. 2006. PMID: 16520384 Free PMC article.
-
Role of Plasmodium falciparum Protein GEXP07 in Maurer's Cleft Morphology, Knob Architecture, and P. falciparum EMP1 Trafficking.mBio. 2020 Mar 17;11(2):e03320-19. doi: 10.1128/mBio.03320-19. mBio. 2020. PMID: 32184257 Free PMC article.
-
Pfsbp1, a Maurer's cleft Plasmodium falciparum protein, is associated with the erythrocyte skeleton.Mol Biochem Parasitol. 2000 Nov;111(1):107-21. doi: 10.1016/s0166-6851(00)00301-7. Mol Biochem Parasitol. 2000. PMID: 11087921
-
The role of the Maurer's clefts in protein transport in Plasmodium falciparum.Trends Parasitol. 2009 Jun;25(6):277-84. doi: 10.1016/j.pt.2009.03.009. Epub 2009 May 11. Trends Parasitol. 2009. PMID: 19442584 Review.
-
Deciphering the export pathway of malaria surface proteins.Trends Parasitol. 2006 Sep;22(9):401-4. doi: 10.1016/j.pt.2006.07.002. Epub 2006 Jul 14. Trends Parasitol. 2006. PMID: 16843728 Review.
Cited by
-
The Plasmodium falciparum transcriptome in severe malaria reveals altered expression of genes involved in important processes including surface antigen-encoding var genes.PLoS Biol. 2018 Mar 12;16(3):e2004328. doi: 10.1371/journal.pbio.2004328. eCollection 2018 Mar. PLoS Biol. 2018. PMID: 29529020 Free PMC article.
-
Repetitive sequences in malaria parasite proteins.FEMS Microbiol Rev. 2017 Nov 1;41(6):923-940. doi: 10.1093/femsre/fux046. FEMS Microbiol Rev. 2017. PMID: 29077880 Free PMC article. Review.
-
Molecular Players at the Sorting Stations of Malaria Parasite 'Plasmodium falciparum'.Curr Protein Pept Sci. 2024;25(6):427-437. doi: 10.2174/0113892037282522240130090156. Curr Protein Pept Sci. 2024. PMID: 38409726 Review.
-
Syk inhibitors interfere with erythrocyte membrane modification during P falciparum growth and suppress parasite egress.Blood. 2017 Aug 24;130(8):1031-1040. doi: 10.1182/blood-2016-11-748053. Epub 2017 Jun 20. Blood. 2017. PMID: 28634183 Free PMC article.
-
Plasmodium knowlesi Skeleton-Binding Protein 1 Localizes to the 'Sinton and Mulligan' Stipplings in the Cytoplasm of Monkey and Human Erythrocytes.PLoS One. 2016 Oct 12;11(10):e0164272. doi: 10.1371/journal.pone.0164272. eCollection 2016. PLoS One. 2016. PMID: 27732628 Free PMC article.
References
-
- Murray CJ, Rosenfeld LC, Lim SS, Andrews KG, Foreman KJ, Haring D, Fullman N, Naghavi M, Lozano R, Lopez AD. Global malaria mortality between 1980 and 2010: a systematic analysis. Lancet. 2012;379:413–431. - PubMed
-
- Cooke BM, Mohandas N, Coppel RL. Malaria and the red blood cell membrane. Semin. Hematol. 2004;41:173–188. - PubMed
-
- Maier AG, Cooke BM, Cowman AF, Tilley L. Malaria parasite proteins that remodel the host erythrocyte. Nat. Rev. Microbiol. 2009;7:341–354. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

