Erythropoietin in traumatic brain injury: study protocol for a randomised controlled trial
- PMID: 25884605
- PMCID: PMC4326467
- DOI: 10.1186/s13063-014-0528-6
Erythropoietin in traumatic brain injury: study protocol for a randomised controlled trial
Abstract
Background: Traumatic brain injury is a leading cause of death and disability worldwide. Laboratory and clinical studies demonstrate a possible beneficial effect of erythropoietin in improving outcomes in the traumatic brain injury cohort. However, there are concerns regarding the association of erythropoietin and thrombosis in the critically ill. A large-scale, multi-centre, blinded, parallel-group, placebo-controlled, randomised trial is currently underway to address this hypothesis.
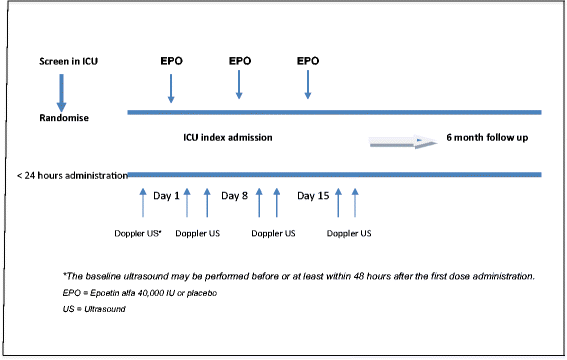
Methods/design: The erythropoietin in traumatic brain injury trial is a stratified prospective, multi-centre, randomised, blinded, parallel-group, placebo-controlled phase III trial. It aims to determine whether the administration of erythropoietin compared to placebo improves neurological outcome in patients with moderate or severe traumatic brain injury at six months after injury. The trial is designed to recruit 606 patients between 15 and 65 years of age with severe (Glasgow Coma Score: 3 to 8) or moderate (Glasgow Coma Score: 9 to 12) traumatic brain injury in Australia, New Zealand, Kingdom of Saudi Arabia, France, Finland, Germany and Ireland. Trial patients will receive either subcutaneous erythropoietin or placebo within 24 hours of injury, and weekly thereafter for up to three doses during the intensive care unit admission. The primary outcome will be the combined proportion of unfavourable neurological outcomes at six months: severe disability or death. Secondary outcomes will include the rate of proximal deep venous thrombosis detected by compression Doppler ultrasound, six-month mortality, the proportion of patients with composite vascular events (deep venous thrombosis, pulmonary embolism, myocardial infarction, cardiac arrest and cerebrovascular events) at six months and quality of life with health economic evaluations.
Discussion: When completed, the trial aims to provide evidence on the efficacy and safety of erythropoietin in traumatic brain injury patients, and to provide clear guidance for clinicians in their management of this devastating condition.
Trial registration: Australian New Zealand Clinical Trials registry: ACTRN12609000827235 (registered on 22 September 2009). Clinicaltrials.gov: NCT00987454 (registered on 29 September 2009). European Drug Regulatory Authorities Clinical Trials: 2011-005235-22 (registered on 18 January 2012).
Figures

References
-
- Access Economics. The economic cost of spinal cord injury and traumatic brain injury in Australia. Report by Access Economics Pty Limited for The Victorian Neurotrauma Initiative. 2009; last access date 28 Jan 2015. http://www.tac.vic.gov.au/about-the-tac/our-organisation/research/tac-ne....
-
- Yatsiv I, Grigoriadis N, Simeonidou C, Stahel PF, Schmidt OI, Alexandrovitch AG, et al. Erythropoietin is neuroprotective, improves functional recovery, and reduces neuronal apoptosis and inflammation in a rodent model of experimental closed head injury. FASEB J. 2005;19:1701–3. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

