Pericytes contribute to the disruption of the cerebral endothelial barrier via increasing VEGF expression: implications for stroke
- PMID: 25884837
- PMCID: PMC4401453
- DOI: 10.1371/journal.pone.0124362
Pericytes contribute to the disruption of the cerebral endothelial barrier via increasing VEGF expression: implications for stroke
Abstract
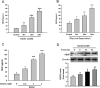
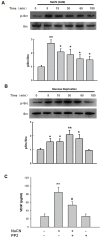
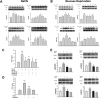
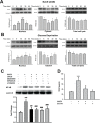
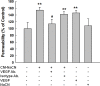
Disruption of the blood-brain barrier (BBB) integrity occurring during the early onset of stroke is not only a consequence of, but also contributes to the further progression of stroke. Although it has been well documented that brain microvascular endothelial cells and astrocytes play a critical role in the maintenance of BBB integrity, pericytes, sandwiched between endothelial cells and astrocytes, remain poorly studied in the pathogenesis of stroke. Our findings demonstrated that treatment of human brain microvascular pericytes with sodium cyanide (NaCN) and glucose deprivation resulted in increased expression of vascular endothelial growth factor (VEGF) via the activation of tyrosine kinase Src, with downstream activation of mitogen activated protein kinase and PI3K/Akt pathways and subsequent translocation of NF-κB into the nucleus. Conditioned medium from NaCN-treated pericytes led to increased permeability of endothelial cells, and this effect was significantly inhibited by VEGF-neutralizing antibody. The in vivo relevance of these findings was further corroborated in the stroke model of mice wherein the mice, demonstrated disruption of the BBB integrity and concomitant increase in the expression of VEGF in the brain tissue as well as in the isolated microvessel. These findings thus suggest the role of pericyte-derived VEGF in modulating increased permeability of BBB during stroke. Understanding the regulation of VEGF expression could open new avenues for the development of potential therapeutic targets for stroke and other neurological disease.
Conflict of interest statement
Figures


References
-
- Zlokovic BV. Cerebrovascular permeability to peptides: manipulations of transport systems at the blood-brain barrier. Pharm Res.1995;12: 1395–1406. - PubMed
-
- Zlokovic BV, Apuzzo ML. Cellular and molecular neurosurgery: pathways from concept to reality—part I: target disorders and concept approaches to gene therapy of the central nervous system. Neurosurgery.1997;40: 789–803. - PubMed
-
- Zlokovic BV, Lipovac MN, Begley DJ, Davson H, Rakic L. Transport of leucine-enkephalin across the blood-brain barrier in the perfused guinea pig brain. J Neurochem.1987;49: 310–315. - PubMed
-
- Zlokovic BV, Begley DJ, Chain-Eliash DG. Blood-brain barrier permeability to leucine-enkephalin, D-alanine2-D-leucine5-enkephalin and their N-terminal amino acid (tyrosine). Brain Res.1985;336: 125–132. - PubMed
-
- Zlokovic BV, Hyman S, McComb JG, Lipovac MN, Tang G, Davson H. Kinetics of arginine-vasopressin uptake at the blood-brain barrier. Biochim Biophys Acta.1990;1025: 191–198. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

