Critical role for IL-18 in spontaneous lung inflammation caused by autophagy deficiency
- PMID: 25888640
- PMCID: PMC4433854
- DOI: 10.4049/jimmunol.1402277
Critical role for IL-18 in spontaneous lung inflammation caused by autophagy deficiency
Abstract
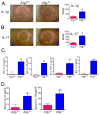
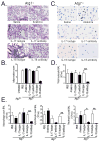
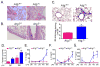
Autophagy is an important component of the immune response. However, the functions of autophagy in human diseases are much less understood. We studied biological consequences of autophagy deficiency in mice lacking the essential autophagy gene Atg7 or Atg5 in myeloid cells. Surprisingly, these mice presented with spontaneous sterile lung inflammation, characterized by marked recruitment of inflammatory cells, submucosal thickening, goblet cell metaplasia, and increased collagen content. Lung inflammation was associated with increase in several proinflammatory cytokines in the bronchoalveolar lavage and in serum. This inflammation was largely driven by IL-18 as a result of constitutive inflammasome activation. Following i.p. LPS injection, autophagy-deficient mice had higher levels of proinflammatory cytokines in lungs and in serum, as well as increased mortality, than control mice. Intranasal bleomycin challenge exacerbated lung inflammation in autophagy-deficient mice and produced more severe fibrotic changes than in control mice. These results uncover a new and important role for autophagy as negative regulator of lung inflammation.
Copyright © 2015 by The American Association of Immunologists, Inc.
Figures




References
-
- Mizushima N, Komatsu M. Autophagy: renovation of cells and tissues. Cell. 2011;147:728–741. - PubMed
-
- Saitoh T, Fujita N, Jang MH, Uematsu S, Yang BG, Satoh T, Omori H, Noda T, Yamamoto N, Komatsu M, Tanaka K, Kawai T, Tsujimura T, Takeuchi O, Yoshimori T, Akira S. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1beta production. Nature. 2008;456:264–268. - PubMed
-
- Kuma A, Hatano M, Matsui M, Yamamoto A, Nakaya H, Yoshimori T, Ohsumi Y, Tokuhisa T, Mizushima N. The role of autophagy during the early neonatal starvation period. Nature. 2004;432:1032–1036. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

