Identification and characterization of epithelial cells derived from human ovarian follicular fluid
- PMID: 25889077
- PMCID: PMC4392788
- DOI: 10.1186/s13287-015-0004-6
Identification and characterization of epithelial cells derived from human ovarian follicular fluid
Abstract
Introduction: Follicular fluid is important for follicular development and oocyte maturation. Evidence suggests that follicular fluid is not only rich in proteins but cells. Besides oocytes, the follicular fluid contains granulosa, thecal, and ovarian surface epithelial cells, and both granulosa and thecal cells are well-characterized. However, epithelial cells in follicular fluid are poorly studied. This study aims to isolate and characterize in vitro epithelial cells that originate from human ovarian follicular fluid retrieved in the assisted fertilization program.
Methods: Follicular fluid samples were collected from 20 women in the assisted reproduction program. Epithelial cells were characterized by flow cytometry assay, immunofluorescence staining, real-time PCR, and time lapse photography.
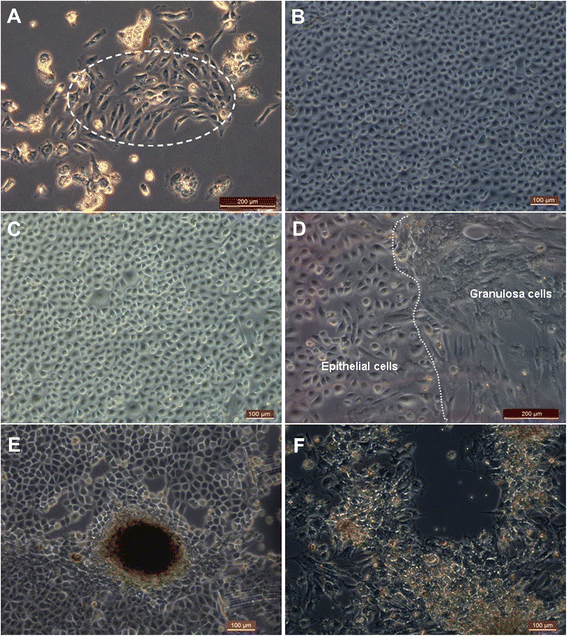
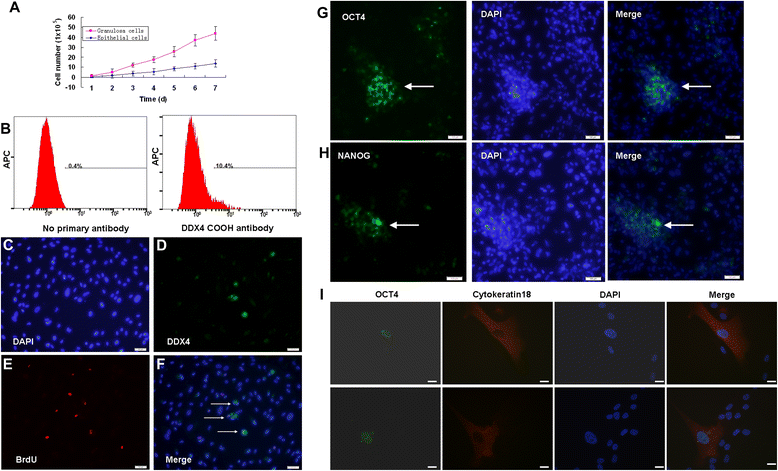
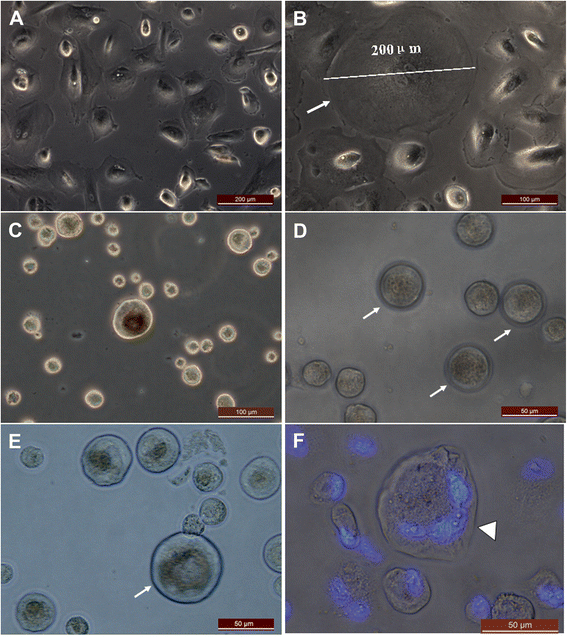
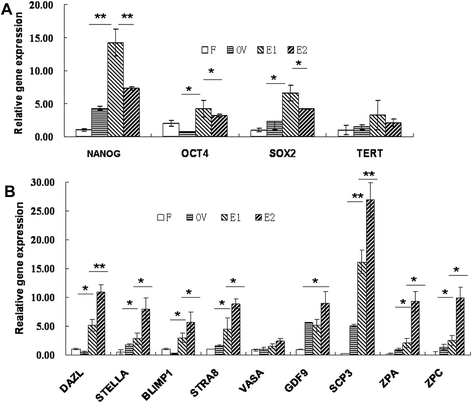
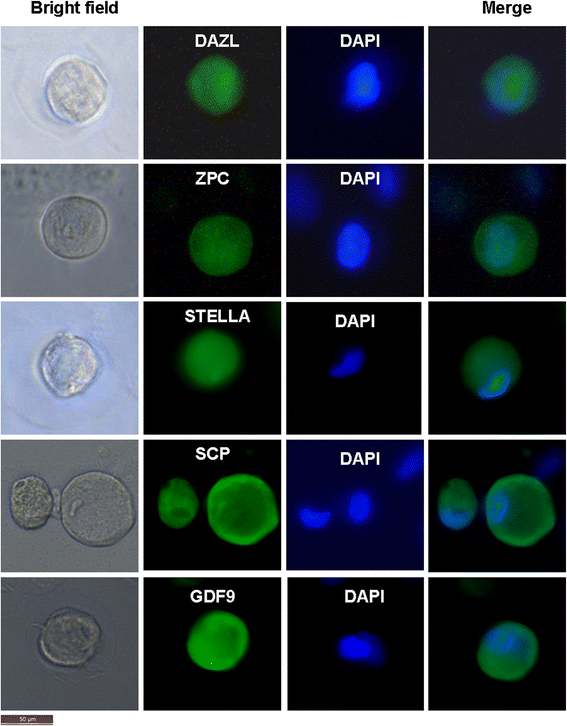
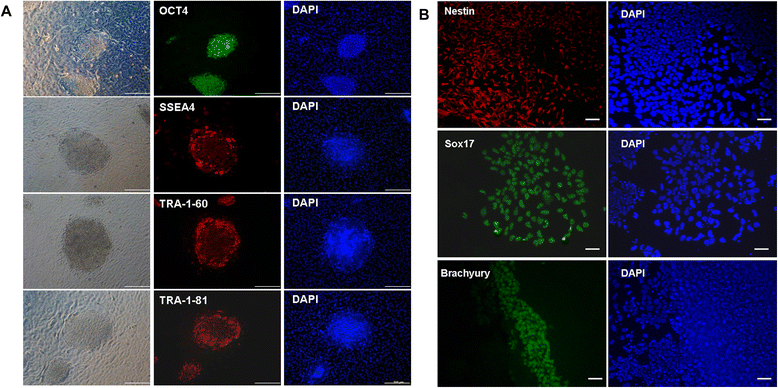
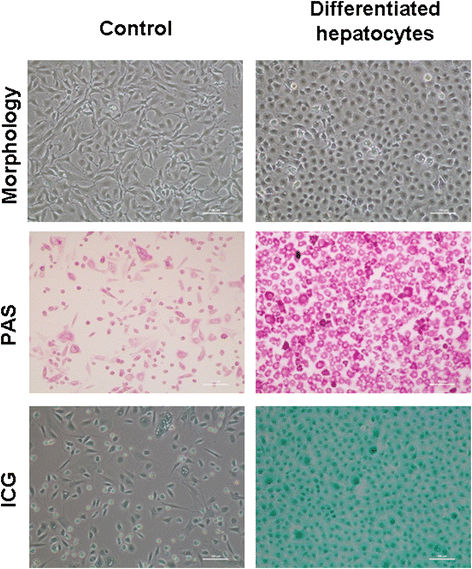
Results: Epithelial cell cultures were established from 18 samples. A small population of epithelial cells expresses germ-line stem cell markers, such as octamer-binding transcription factor 4 (OCT4), NANOG, and DEAD box polypeptide 4 (DDX4). In the epithelial cell culture system, oocyte-like cells formed spontaneously in vitro and expressed the following transcription markers: deleted in azoospermia-like (DAZL), developmental pluripotency associated protein 3 stella-related protein (STELLA), zona pellucida gene family C (ZPC), Syntaptonemal complex protein (SCP), and growth and differentiation factor 9 (GDF9). Some of the oocyte-like cells developed a zona pellucida-like structure. Both the symmetric and asymmetric division split of epithelial cells and early developing oocytes were observed using time lapse photography. Cell colonies were formed during epithelial culturing, which maintained and proliferated in an undifferentiated way on the feeder layer and expressed some pluripotency markers. These colonies differentiated in vitro into various somatic cell types in all three germ layers, but did not form teratoma when injected into immunodeficient mice. Furthermore, these epithelial cells could be differentiated directly to functional hepatocyte-like cells, which do not exist in ovarian tissues.
Conclusions: The epithelial cells derived from follicular fluid are a potential stem cell source with a pluripotent/multipotent character for safe application in oogenesis and regenerative medicine.
Figures

Similar articles
-
Retinoic acid induces mouse bone marrow-derived CD15⁺, Oct4⁺ and CXCR4⁺ stem cells into male germ-like cells in a two-dimensional cell culture system.Cell Biol Int. 2014 Jun;38(6):782-9. doi: 10.1002/cbin.10260. Epub 2014 Mar 14. Cell Biol Int. 2014. PMID: 24677291
-
Induction of E-cadherin+ human amniotic fluid cell differentiation into oocyte-like cells via culture in medium supplemented with follicular fluid.Mol Med Rep. 2014 Jul;10(1):21-8. doi: 10.3892/mmr.2014.2199. Epub 2014 Apr 29. Mol Med Rep. 2014. PMID: 24788191 Free PMC article.
-
Developmental and molecular aberrations associated with deterioration of oogenesis during complete or partial follicle-stimulating hormone receptor deficiency in mice.Biol Reprod. 2003 Oct;69(4):1294-302. doi: 10.1095/biolreprod.103.015610. Epub 2003 Jun 11. Biol Reprod. 2003. PMID: 12801992
-
Features that affect secretion and assembly of zona pellucida glycoproteins during mammalian oogenesis.Soc Reprod Fertil Suppl. 2007;63:187-201. Soc Reprod Fertil Suppl. 2007. PMID: 17566273 Review.
-
Structural, biochemical and functional aspects of sperm-oocyte interactions in pigs.Soc Reprod Fertil Suppl. 2006;62:317-30. Soc Reprod Fertil Suppl. 2006. PMID: 16866327 Review.
Cited by
-
Epigenetic age acceleration in follicular fluid and markers of ovarian response among women undergoing IVF.Hum Reprod. 2024 Sep 1;39(9):2003-2009. doi: 10.1093/humrep/deae136. Hum Reprod. 2024. PMID: 38890131 Free PMC article.
-
Female Age Affects the Mesenchymal Stem Cell Characteristics of Aspirated Follicular Cells in the In Vitro Fertilization Programme.Stem Cell Rev Rep. 2019 Aug;15(4):543-557. doi: 10.1007/s12015-019-09889-0. Stem Cell Rev Rep. 2019. PMID: 31055736 Clinical Trial.
-
Human Ovarian Follicular Fluid Mesenchymal Stem Cells Express Osteogenic Markers When Cultured on Bioglass 58S-Coated Titanium Scaffolds.Materials (Basel). 2023 May 11;16(10):3676. doi: 10.3390/ma16103676. Materials (Basel). 2023. PMID: 37241304 Free PMC article.
-
Utilizing Calcium Alginate for the Assessment of Bone Morphogenetic Protein 15 Induction Effect on the Differentiation of Mesenchymal Stem Cell Derived from Human Follicular Fluid to Oocyte-Like Structure.Adv Biomed Res. 2020 Dec 23;9:80. doi: 10.4103/abr.abr_200_20. eCollection 2020. Adv Biomed Res. 2020. PMID: 33912496 Free PMC article.
-
A primary insight into gut microbiome, MicroRNA and stemness, in a PCOS rat model.J Ovarian Res. 2025 Apr 1;18(1):66. doi: 10.1186/s13048-025-01648-9. J Ovarian Res. 2025. PMID: 40170042 Free PMC article.
References
-
- Auersperg N, Wong AS, Choi KC, Kang SK, Leung PC. Ovarian surface epithelium: biology, endocrinology, and pathology. Endocr Rev. 2001;22:255–88. - PubMed
-
- Brůcková L, Soukup T, Moos J, Moosová M, Pavelková J, Rezábek K, et al. The cultivation of human granulosa cells. Acta Med (Hradec Kralove). 2008;51:165–72. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

