Splenic architecture disruption and parasite-induced splenocyte activation and anergy in Plasmodium falciparum-infected Saimiri sciureus monkeys
- PMID: 25890318
- PMCID: PMC4377215
- DOI: 10.1186/s12936-015-0641-3
Splenic architecture disruption and parasite-induced splenocyte activation and anergy in Plasmodium falciparum-infected Saimiri sciureus monkeys
Abstract
Background: The understanding of the mechanisms of immunity in malaria is crucial for the rational development of interventions such as vaccines. During blood stage infection, the spleen is considered to play critical roles in both immunity and immunopathology of Plasmodium falciparum infections.
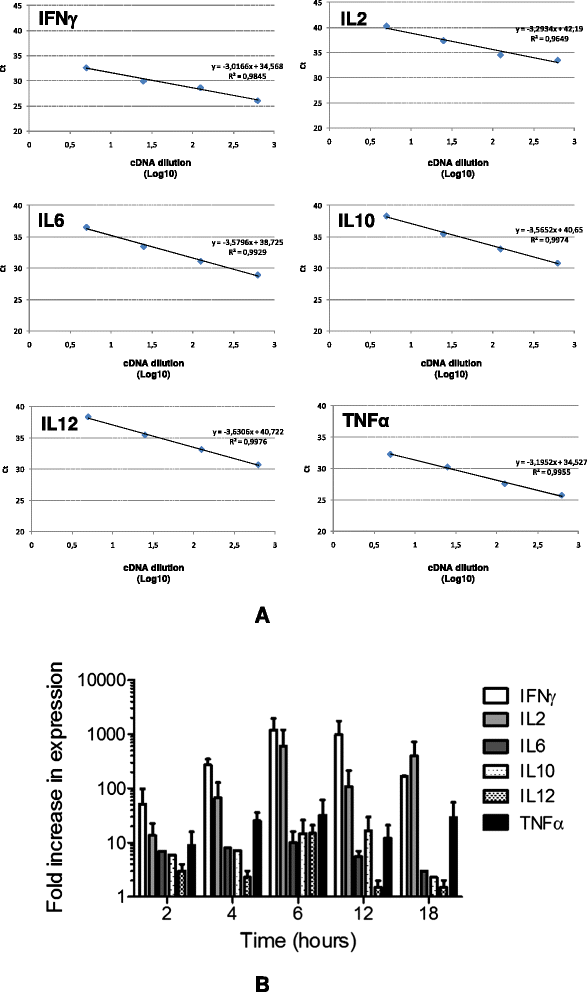
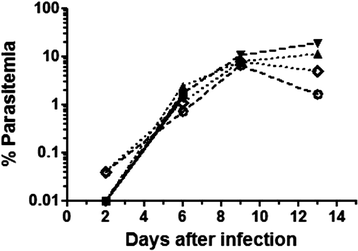
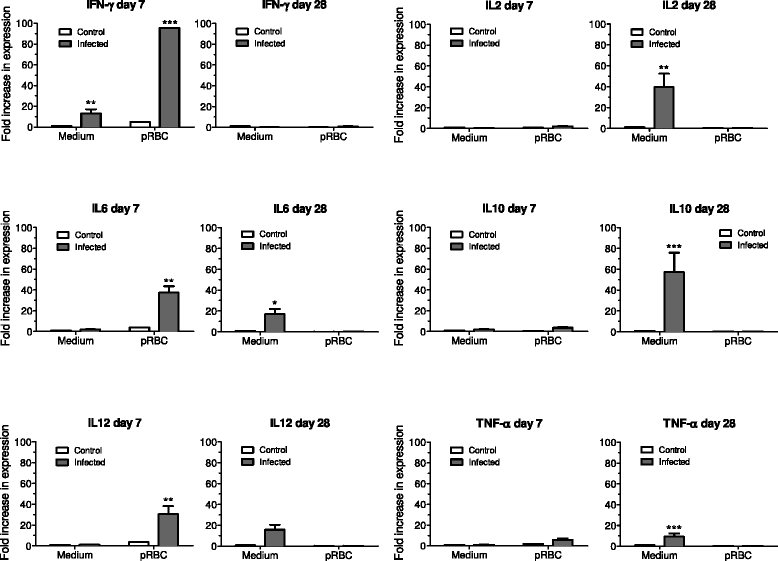
Methods: Saimiri sciureus monkeys were inoculated with blood stages of P. falciparum (FUP strain) and spleens removed during acute disease (days 7 and 13 of infection) and during convalescence (15 days after start of chloroquine treatment). Cytokine (IFNγ, TNFα, IL2, IL6, IL10, and IL12) responses of splenocytes stimulated with P. falciparum-parasitized red blood cells were assessed by real-time PCR using specific Saimiri primers, and histological changes were evaluated using haematoxylin-eosin and Giemsa-stained slides.
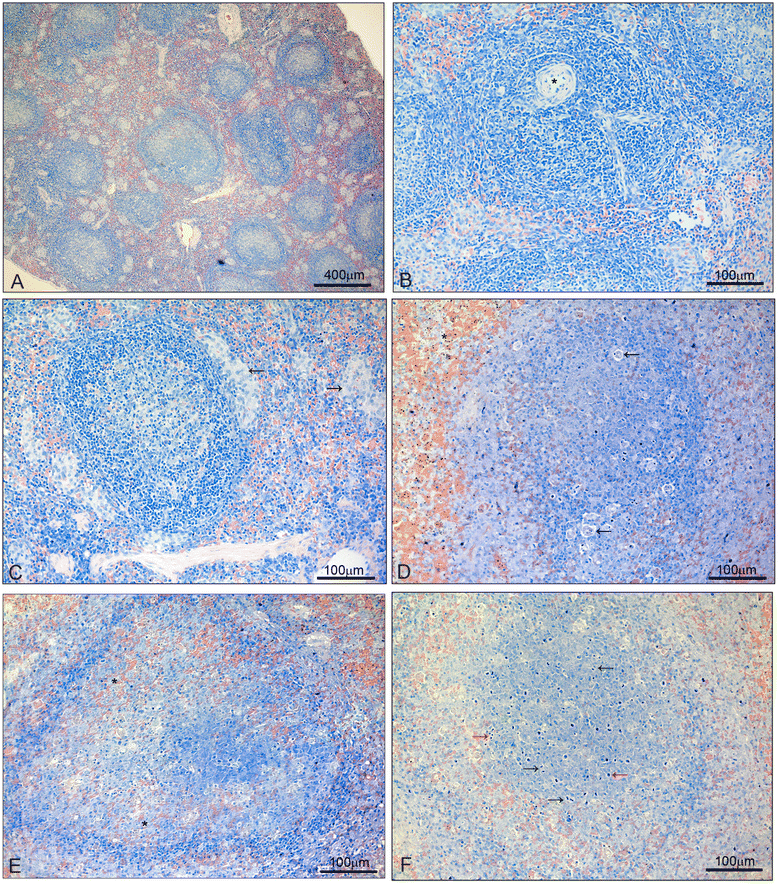
Results: Early during infection (day 7, 1-2% parasitaemia), spleens showed disruption of germinal centre architecture with heavy B-cell activation (centroblasts), and splenocytes showed increased expression of IFNγ, IL6 and IL12 upon in vitro stimuli by P. falciparum-parasitized red blood cells (pRBC). Conversely, 15 days after treatment of blood stage infection with chloroquine, splenocytes showed spontaneous in vitro expression of TNFα, IL2, IL6, IL10, and IL12, but not IFNγ, and stimulation with P. falciparum pRBC blocked the expression of all these cytokines. During the acute phase of infection, splenic disarray with disorganized germinal centres was observed. During convalescence, spleens of the chloroquine-treated animals showed white pulp hyperplasia with extensive lymphocyte activation and persistency of heavily haemozoin-laden macrophages throughout the red pulp.
Conclusions: Inability to eliminate haemozoin is likely involved in the persistent lymphocyte activation and in the anergic responses of Saimiri splenocytes to P. falciparum pRBC, with important negative impact in immune responses and implications for the design of malaria vaccine.
Figures

References
-
- Baird JK, Sismadi P, Masbar S, Ramzan A, Purnomo BW, Sekartuti A, et al. A focus of endemic malaria in Central Java. Am J Trop Med Hyg. 1996;54:98–104. - PubMed
-
- Baird JK, Agyei SO, Utz GC, Koram K, Barcus MJ, Jones TR, et al. Seasonal malaria attack rates in infants and young children in northen Ghana. Am J Trop Med Hyg. 2002;66:280–6. - PubMed
-
- Perlmann P, Troye-Blomberg M. Malaria immunology. Folia Parasitol. 2002;49:304. doi: 10.14411/fp.2002.056. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

