Alzheimer's disease pathology is attenuated in a CD38-deficient mouse model
- PMID: 25893674
- PMCID: PMC6929692
- DOI: 10.1002/ana.24425
Alzheimer's disease pathology is attenuated in a CD38-deficient mouse model
Abstract
Objective: Alzheimer's disease (AD)-associated dementia is due to tissue damage caused by amyloid β (Aβ) deposition within the brain and by accompanying neuroinflammation. The nicotinamide adenine dinucleotide (NAD) glycohydrolase CD38, which is expressed by neurons, astrocytes, and microglial cells, regulates inflammatory and repair processes in the brain and other tissues by degrading NAD and repressing the activity of other NAD-consuming enzymes and by producing NAD-derived metabolites that regulate calcium signaling and migration of inflammatory cells. Given the role of CD38 in neuroinflammation and repair, we examined the effect of CD38 deletion on AD pathology.
Methods: We crossed APPswePS1ΔE9 (APP.PS) mice with Cd38(-) (/) (-) mice to generate AD-prone CD38-deficient animals (APP.PS.Cd38(-) (/) (-) ) and examined AD-related phenotypes in both groups.
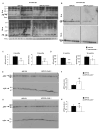
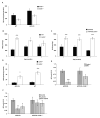
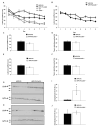
Results: APP.PS.Cd38(-) (/) (-) mice exhibited significant reductions in Aβ plaque load and soluble Aβ levels compared to APP.PS mice, and this correlated with improved spatial learning. Although CD38 deficiency resulted in decreased microglia/macrophage (MM) accumulation, the transcription profile of the Cd38(-) (/) (-) and Cd38(+/) (+) MM was similar, suggesting that the decreased Aβ burden in APP.PS.Cd38(-) (/) (-) mice was not due to alterations in MM activation/function. Instead, APP.PS.Cd38(-) (/) (-) neuronal cultures secreted less Aβ and this reduction was mimicked when APP.PS neuronal cultures were treated with inhibitors that blocked CD38 enzyme activity or the signaling pathways controlled by CD38-derived metabolites. Furthermore, β- and γ-secretase activity was decreased in APP.PS.Cd38(-) (/) (-) mice, which correlated with decreased Aβ production.
Interpretation: CD38 regulates AD pathology in the APP.PS model of AD, suggesting that CD38 may be a novel target for AD treatment.
© 2015 American Neurological Association.
Conflict of interest statement
R.S., A.L., E.B., and A.B.,: grant, Teva
F.L., None declared
T.D., None declared
T.H., None declared
M.G., None declared
V.H., None declared
Figures




References
-
- Itagaki S, McGeer PL, Akiyama H, et al. Relationship of microglia and astrocytes to amyloid deposits of Alzheimer disease. Journal of neuroimmunology. 1989;24:173–182. - PubMed
-
- El Khoury J, Toft M, Hickman SE, et al. Ccr2 deficiency impairs microglial accumulation and accelerates progression of Alzheimer-like disease. Nature medicine. 2007;13:432–438. - PubMed
-
- Kettenmann H, Hanisch UK, Noda M, et al. Physiology of microglia. Physiological reviews. 2011;91:461–553. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

