The Tissue Fibrinolytic System Contributes to the Induction of Macrophage Function and CCL3 during Bone Repair in Mice
- PMID: 25893677
- PMCID: PMC4404328
- DOI: 10.1371/journal.pone.0123982
The Tissue Fibrinolytic System Contributes to the Induction of Macrophage Function and CCL3 during Bone Repair in Mice
Abstract
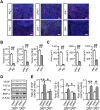
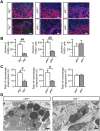
Macrophages play crucial roles in repair process of various tissues. However, the details in the role of macrophages during bone repair still remains unknown. Herein, we examined the contribution of the tissue fibrinolytic system to the macrophage functions in bone repair after femoral bone defect by using male mice deficient in plasminogen (Plg-/-), urokinase-type plasminogen activator (uPA-/-) or tissue-type plasminogen activator (tPA-/-) genes and their wild-type littermates. Bone repair of the femur was delayed in uPA-/- mice until day 6, compared with wild-type (uPA+/+) mice. Number of Osterix-positive cells and vessel formation were decreased in uPA-/- mice at the bone injury site on day 4, compared with those in uPA+/+ mice. Number of macrophages and their phagocytosis at the bone injury site were reduced in uPA-/- and Plg-/-, but not in tPA-/- mice on day 4. Although uPA or plasminogen deficiency did not affect the levels of cytokines, including TNF-α, IL-1β, IL-6, IL-4 and IFN-γ mRNA in the damaged femur, the elevation in CCL3 mRNA levels was suppressed in uPA-/- and Plg-/-, but not in tPA-/- mice. Neutralization of CCL3 antagonized macrophage recruitment to the site of bone injury and delayed bone repair in uPA+/+, but not in uPA-/- mice. Our results provide novel evidence that the tissue fibrinolytic system contributes to the induction of macrophage recruitment and CCL3 at the bone injury site, thereby, leading to the enhancement of the repair process.
Conflict of interest statement
Figures





Similar articles
-
Tissue-type plasminogen activator deficiency delays bone repair: roles of osteoblastic proliferation and vascular endothelial growth factor.Am J Physiol Endocrinol Metab. 2014 Aug 1;307(3):E278-88. doi: 10.1152/ajpendo.00129.2014. Epub 2014 Jun 10. Am J Physiol Endocrinol Metab. 2014. PMID: 24918201
-
Plasminogen activation independent of uPA and tPA maintains wound healing in gene-deficient mice.EMBO J. 2006 Jun 21;25(12):2686-97. doi: 10.1038/sj.emboj.7601173. Epub 2006 Jun 8. EMBO J. 2006. PMID: 16763560 Free PMC article.
-
Increased bone formation in mice lacking plasminogen activators.J Bone Miner Res. 2003 Jul;18(7):1167-76. doi: 10.1359/jbmr.2003.18.7.1167. J Bone Miner Res. 2003. PMID: 12854826
-
The Roles of Fibrinolytic Factors in Bone Destruction Caused by Inflammation.Cells. 2024 Mar 15;13(6):516. doi: 10.3390/cells13060516. Cells. 2024. PMID: 38534360 Free PMC article. Review.
-
Roles of fibrinolytic factors in the alterations in bone marrow hematopoietic stem/progenitor cells during bone repair.Inflamm Regen. 2020 Sep 16;40:22. doi: 10.1186/s41232-020-00128-5. eCollection 2020. Inflamm Regen. 2020. PMID: 32944096 Free PMC article. Review.
Cited by
-
Hdac3 deficiency limits periosteal reaction associated with Western diet feeding in female mice.J Cell Mol Med. 2024 Sep;28(17):e70081. doi: 10.1111/jcmm.70081. J Cell Mol Med. 2024. PMID: 39261913 Free PMC article.
-
Role of Macrophages and Plasminogen Activator Inhibitor-1 in Delayed Bone Repair Induced by Glucocorticoids in Mice.Int J Mol Sci. 2022 Jan 1;23(1):478. doi: 10.3390/ijms23010478. Int J Mol Sci. 2022. PMID: 35008904 Free PMC article.
-
Shear and Dynamic Compression Modulates the Inflammatory Phenotype of Human Monocytes in vitro.Front Immunol. 2019 Mar 5;10:383. doi: 10.3389/fimmu.2019.00383. eCollection 2019. Front Immunol. 2019. PMID: 30891042 Free PMC article.
-
Macrophage Transplantation Fails to Improve Repair of Critical-Sized Calvarial Defects.J Craniofac Surg. 2019 Nov-Dec;30(8):2640-2645. doi: 10.1097/SCS.0000000000005797. J Craniofac Surg. 2019. PMID: 31609958 Free PMC article.
-
Roles of plasminogen in the alterations in bone marrow hematopoietic stem cells during bone repair.Bone Rep. 2018 Apr 27;8:195-203. doi: 10.1016/j.bonr.2018.04.005. eCollection 2018 Jun. Bone Rep. 2018. PMID: 29955638 Free PMC article.
References
-
- Singer AJ, Clark RA. Cutaneous wound healing. N Engl J Med. 1999;341: 738–746. - PubMed
-
- Maurer M, von Stebut E. Macrophage inflammatory protein-1. Int J Biochem Cell Biol. 2004;36: 1882–1886. - PubMed
-
- Gordon S. Alternative activation of macrophages. Nat Rev Immunol. 2003;3: 23–35. - PubMed
-
- Castellino FJ, Ploplis VA. Structure and function of the plasminogen/plasmin system. Thromb Haemost. 2005;93: 647–654. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

