Anti-Pseudomonas aeruginosa IgY Antibodies Induce Specific Bacterial Aggregation and Internalization in Human Polymorphonuclear Neutrophils
- PMID: 25895968
- PMCID: PMC4468541
- DOI: 10.1128/IAI.02970-14
Anti-Pseudomonas aeruginosa IgY Antibodies Induce Specific Bacterial Aggregation and Internalization in Human Polymorphonuclear Neutrophils
Abstract
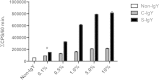
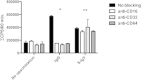
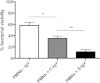
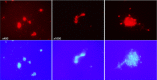
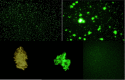
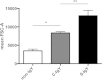
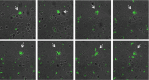
Polymorphonuclear neutrophils (PMNs) are essential cellular constituents in the innate host response, and their recruitment to the lungs and subsequent ubiquitous phagocytosis controls primary respiratory infection. Cystic fibrosis pulmonary disease is characterized by progressive pulmonary decline governed by a persistent, exaggerated inflammatory response dominated by PMNs. The principal contributor is chronic Pseudomonas aeruginosa biofilm infection, which attracts and activates PMNs and thereby is responsible for the continuing inflammation. Strategies to prevent initial airway colonization with P. aeruginosa by augmenting the phagocytic competence of PMNs may postpone the deteriorating chronic biofilm infection. Anti-P. aeruginosa IgY antibodies significantly increase the PMN-mediated respiratory burst and subsequent bacterial killing of P. aeruginosa in vitro. The mode of action is attributed to IgY-facilitated formation of immobilized bacteria in aggregates, as visualized by fluorescence microscopy and the induction of increased bacterial hydrophobicity. Thus, the present study demonstrates that avian egg yolk immunoglobulins (IgY) targeting P. aeruginosa modify bacterial fitness, which enhances bacterial killing by PMN-mediated phagocytosis and thereby may facilitate a rapid bacterial clearance in airways of people with cystic fibrosis.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures

Similar articles
-
Anti-Pseudomonas aeruginosa IgY antibodies promote bacterial opsonization and augment the phagocytic activity of polymorphonuclear neutrophils.Hum Vaccin Immunother. 2016 Jul 2;12(7):1690-9. doi: 10.1080/21645515.2016.1145848. Epub 2016 Feb 22. Hum Vaccin Immunother. 2016. PMID: 26901841 Free PMC article.
-
Anti-Pseudomonas aeruginosa IgY antibodies augment bacterial clearance in a murine pneumonia model.J Cyst Fibros. 2016 Mar;15(2):171-8. doi: 10.1016/j.jcf.2015.08.002. Epub 2015 Aug 21. J Cyst Fibros. 2016. PMID: 26303991
-
Azithromycin potentiates avian IgY effect against Pseudomonas aeruginosa in a murine pulmonary infection model.Int J Antimicrob Agents. 2021 Jan;57(1):106213. doi: 10.1016/j.ijantimicag.2020.106213. Epub 2020 Oct 23. Int J Antimicrob Agents. 2021. PMID: 33256950
-
Mechanisms of phagocytosis and host clearance of Pseudomonas aeruginosa.Am J Physiol Lung Cell Mol Physiol. 2014 Apr 1;306(7):L591-603. doi: 10.1152/ajplung.00335.2013. Epub 2014 Jan 24. Am J Physiol Lung Cell Mol Physiol. 2014. PMID: 24464809 Free PMC article. Review.
-
Interactions between Neutrophils and Pseudomonas aeruginosa in Cystic Fibrosis.Pathogens. 2017 Mar 9;6(1):10. doi: 10.3390/pathogens6010010. Pathogens. 2017. PMID: 28282951 Free PMC article. Review.
Cited by
-
Immune Responses to Pseudomonas aeruginosa Biofilm Infections.Front Immunol. 2021 Feb 22;12:625597. doi: 10.3389/fimmu.2021.625597. eCollection 2021. Front Immunol. 2021. PMID: 33692800 Free PMC article. Review.
-
Immunotherapy with IgY Antibodies toward Outer Membrane Protein F Protects Burned Mice against Pseudomonas aeruginosa Infection.J Immunol Res. 2020 May 29;2020:7840631. doi: 10.1155/2020/7840631. eCollection 2020. J Immunol Res. 2020. PMID: 32566689 Free PMC article.
-
Polyclonal anti-whole cell IgY passive immunotherapy shields against P. aeruginosa-induced acute pneumonia and burn wound infections in murine models.Sci Rep. 2024 Jan 3;14(1):405. doi: 10.1038/s41598-023-50859-x. Sci Rep. 2024. PMID: 38172232 Free PMC article.
-
Immunoglobulin Y for Potential Diagnostic and Therapeutic Applications in Infectious Diseases.Front Immunol. 2021 Jun 9;12:696003. doi: 10.3389/fimmu.2021.696003. eCollection 2021. Front Immunol. 2021. PMID: 34177963 Free PMC article. Review.
-
A systematic review of the avian antibody (IgY) therapeutic effects on human bacterial infections over the decade.Antib Ther. 2025 Mar 9;8(2):111-123. doi: 10.1093/abt/tbaf007. eCollection 2025 Apr. Antib Ther. 2025. PMID: 40177645 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

