Stability of Minimum Essential Medium functionality despite L-glutamine decomposition
- PMID: 25904557
- PMCID: PMC4960165
- DOI: 10.1007/s10616-015-9875-8
Stability of Minimum Essential Medium functionality despite L-glutamine decomposition
Abstract
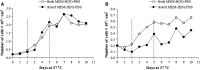
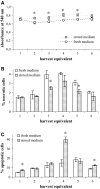
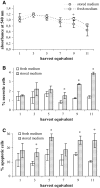
L-Glutamine (L-Gln) instability in liquid media is a well-known fact. Also, negative effect of ammonia, one of the L-Gln degradation products, on viability of many cell cultures and on replication of different viruses has been described. However, negative effects of ammonia have been reported in doses excessively exceeding those that could be generated in regularly used liquid culture media due to spontaneous L-Gln breakdown (below 2 mM). Traditional virus vaccine production processes have been established and registered involving L-Gln containing media use. Eventual culture media replacement in the regular production process belongs to the major regulative changes that require substantial financial expenses. The aim of this study was to evaluate the effect of storage of Minimum Essential Media with Hanks salts on their relevant biological functions during virus vaccine production process in relation to L-Gln decrease. Our results show a cell type dependent effect of spontaneous L-Gln degradation during medium storage. They also suggest that for cell cultures used in measles, mumps, and rubella virus production the media retain their functionality in respect to cell viability or virus growth over a certain time window despite L-Gln degradation.
Keywords: Apoptosis; Cell viability; L-Glutamine; Measles; Minimum Essential Medium (MEM); Mumps; Necrosis; Rubella.
Figures




Similar articles
-
Development of a serum-free and heat-sterilizable medium and continuous high-density cell culture.Cytotechnology. 1991;5 Suppl 2:S35-51. Cytotechnology. 1991. PMID: 1367253 Review.
-
Influence of glutamine-supplemented Caco-2 cells on cytokine production of mononuclear cells.JPEN J Parenter Enteral Nutr. 2003 Sep-Oct;27(5):333-9. doi: 10.1177/0148607103027005333. JPEN J Parenter Enteral Nutr. 2003. PMID: 12971733
-
Glutamine is essential for nitric oxide synthesis by murine macrophages.J Surg Res. 1999 Oct;86(2):213-9. doi: 10.1006/jsre.1999.5713. J Surg Res. 1999. PMID: 10534426
-
Influence of two glutamine-containing dipeptides on growth of mammalian cells.In Vitro Cell Dev Biol. 1988 Jul;24(7):696-8. doi: 10.1007/BF02623607. In Vitro Cell Dev Biol. 1988. PMID: 3397369
-
Measles, mumps, and rubella virus vaccine (M-M-R™II): a review of 32 years of clinical and postmarketing experience.Vaccine. 2012 Nov 6;30(48):6918-26. doi: 10.1016/j.vaccine.2012.08.057. Epub 2012 Sep 7. Vaccine. 2012. PMID: 22959986 Review.
Cited by
-
Improving Mechanical Properties of Tendon Allograft through Rehydration Strategies: An In Vitro Study.Bioengineering (Basel). 2023 May 25;10(6):641. doi: 10.3390/bioengineering10060641. Bioengineering (Basel). 2023. PMID: 37370572 Free PMC article.
-
Adeno-associated virus perfusion enhanced expression: A commercially scalable, high titer, high quality producer cell line process.Mol Ther Methods Clin Dev. 2024 May 14;32(2):101266. doi: 10.1016/j.omtm.2024.101266. eCollection 2024 Jun 13. Mol Ther Methods Clin Dev. 2024. PMID: 38868441 Free PMC article.
-
Design considerations of benchtop fluid flow bioreactors for bio-engineered tissue equivalents in vitro.Biomater Biosyst. 2022 Aug 31;8:100063. doi: 10.1016/j.bbiosy.2022.100063. eCollection 2022 Dec. Biomater Biosyst. 2022. PMID: 36824373 Free PMC article.
-
Microfabricated polymer-metal biosensors for multifarious data collection from electrogenic cellular models.Microsyst Nanoeng. 2023 Mar 1;9:22. doi: 10.1038/s41378-023-00488-1. eCollection 2023. Microsyst Nanoeng. 2023. PMID: 36875634 Free PMC article.
-
Current utilization trend of immortalized mast cell lines in allergy research: a systematic review.Immunol Res. 2025 Jan 21;73(1):41. doi: 10.1007/s12026-024-09562-w. Immunol Res. 2025. PMID: 39838115 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

