Acquired coagulant factor VIII deficiency induced by Bacillus anthracis lethal toxin in mice
- PMID: 25906166
- PMCID: PMC4601479
- DOI: 10.1080/21505594.2015.1031454
Acquired coagulant factor VIII deficiency induced by Bacillus anthracis lethal toxin in mice
Abstract
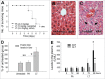
Mice treated with anthrax lethal toxin (LT) exhibit hemorrhage caused by unknown mechanisms. Moreover, LT treatment in mice induced liver damage. In this study, we hypothesized that a suppressed coagulation function may be associated with liver damage, because the liver is the major producing source of coagulation factors. The hepatic expression of coagulant factors and the survival rates were analyzed after cultured cells or mice were exposed to LT. In agreement with our hypothesis, LT induces cytotoxicity against hepatic cells in vitro. In addition, suppressed expression of coagulation factor VIII (FVIII) in the liver is associated with a prolonged plasma clotting time in LT-treated mice, suggesting a suppressive role of LT in coagulation. Accordingly, we further hypothesized that a loss-of-function approach involving treatments of an anticoagulant should exacerbate LT-induced abnormalities, whereas a gain-of-function approach involving injections of recombinant FVIII to complement the coagulation deficiency should ameliorate the pathogenesis. As expected, a sublethal dose of LT caused mortality in the mice that were non-lethally pretreated with an anticoagulant (warfarin). By contrast, treatments of recombinant FVIII reduced the mortality from a lethal dose of LT in mice. Our results indicated that LT-induced deficiency of FVIII is involved in LT-mediated pathogenesis. Using recombinant FVIII to correct the coagulant defect may enable developing a new strategy to treat anthrax.
Keywords: Anthrax; coagulation factor VIII; hemorrhage; lethal toxin.
Figures

References
-
- Dixon TC, Meselson M, Guillemin J, Hanna PC. Anthrax. N Engl J Med 1999; 341:815-26; PMID:10477781; http://dx.doi.org/ 10.1056/NEJM199909093411107 - DOI - PubMed
-
- Collier RJ, Young JA. Anthrax toxin. Annu Rev Cell Dev Biol 2003; 19:45-70; PMID:14570563; http://dx.doi.org/ 10.1146/annurev.cellbio.19.111301.140655 - DOI - PubMed
-
- Moayeri M, Leppla SH. The roles of anthrax toxin in pathogenesis. Curr Opin Microbiol 2004; 7:19-24; PMID:15036135; http://dx.doi.org/ 10.1016/j.mib.2003.12.001 - DOI - PubMed
-
- Kau JH, Shih YL, Lien TS, Lee CC, Huang HH, Lin HC, Sun DS, Chang HH. Activated protein C ameliorates Bacillus anthracis lethal toxin-induced lethal pathogenesis in rats. J Biomed Sci 2012; 19:98; PMID:23170801; http://dx.doi.org/ 10.1186/1423-0127-19-98 - DOI - PMC - PubMed
-
- Kau JH, Sun DS, Huang HH, Wong MS, Lin HC, Chang HH. Role of visible light-activated photocatalyst on the reduction of anthrax spore-induced mortality in mice. PloS One 2009; 4:e4167; PMID:19132100; http://dx.doi.org/ 10.1371/journal.pone.0004167 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
