The involvement of wheat F-box protein gene TaFBA1 in the oxidative stress tolerance of plants
- PMID: 25906259
- PMCID: PMC4408080
- DOI: 10.1371/journal.pone.0122117
The involvement of wheat F-box protein gene TaFBA1 in the oxidative stress tolerance of plants
Abstract
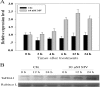
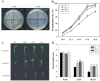
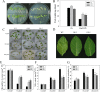
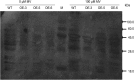
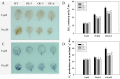
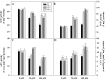
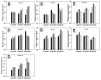
As one of the largest gene families, F-box domain proteins have been found to play important roles in abiotic stress responses via the ubiquitin pathway. TaFBA1 encodes a homologous F-box protein contained in E3 ubiquitin ligases. In our previous study, we found that the overexpression of TaFBA1 enhanced drought tolerance in transgenic plants. To investigate the mechanisms involved, in this study, we investigated the tolerance of the transgenic plants to oxidative stress. Methyl viologen was used to induce oxidative stress conditions. Real-time PCR and western blot analysis revealed that TaFBA1 expression was up-regulated by oxidative stress treatments. Under oxidative stress conditions, the transgenic tobacco plants showed a higher germination rate, higher root length and less growth inhibition than wild type (WT). The enhanced oxidative stress tolerance of the transgenic plants was also indicated by lower reactive oxygen species (ROS) accumulation, malondialdehyde (MDA) content and cell membrane damage under oxidative stress compared with WT. Higher activities of antioxidant enzymes, including superoxide dismutase (SOD), catalase (CAT), ascorbate peroxidase (APX) and peroxidase (POD), were observed in the transgenic plants than those in WT, which may be related to the upregulated expression of some antioxidant genes via the overexpression of TaFBA1. In others, some stress responsive elements were found in the promoter region of TaFBA1, and TaFBA1 was located in the nucleus, cytoplasm and plasma membrane. These results suggest that TaFBA1 plays an important role in the oxidative stress tolerance of plants. This is important for understanding the functions of F-box proteins in plants' tolerance to multiple stress conditions.
Conflict of interest statement
Figures

References
-
- Sharma P, Jha AB, Dubey RS, Pessarakli M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot. 2012; 2012: article ID 217037.
-
- López MA, Vicente J, Kulasekaran S, Vellosillo T, Marti´nez M, Irigoyen ML, et al. Antagonistic role of 9-lipoxygenase-derived oxylipins and ethylene in the control of oxidative stress, lipid peroxidation and plant defence. Plant J. 2011;67(3): 447–458. 10.1111/j.1365-313X.2011.04608.x - DOI - PubMed
-
- Mittler R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002;7: 405–410. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

