Economic evaluation of hormonal therapies for postmenopausal women with estrogen receptor-positive early breast cancer in Canada
- PMID: 25908907
- PMCID: PMC4399616
- DOI: 10.3747/co.22.2120
Economic evaluation of hormonal therapies for postmenopausal women with estrogen receptor-positive early breast cancer in Canada
Abstract
Background: Aromatase inhibitor (ai) therapy has been subjected to numerous cost-effectiveness analyses. However, with most ais having reached the end of patent protection and with maturation of the clinical trials data, a re-analysis of ai cost-effectiveness and a consideration of ai use as part of sequential therapy is desirable. Our objective was to assess the cost-effectiveness of the 5-year upfront and sequential tamoxifen (tam) and ai hormonal strategies currently used for treating patients with estrogen receptor (er)-positive early breast cancer.
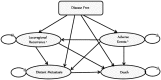
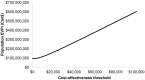
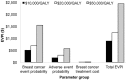
Methods: The cost-effectiveness analysis used a Markov model that took a Canadian health system perspective with a lifetime time horizon. The base case involved 65-year-old women with er-positive early breast cancer. Probabilistic sensitivity analyses were used to incorporate parameter uncertainties. An expected-value-of-perfect-information test was performed to identify future research directions. Outcomes were quality-adjusted life-years (qalys) and costs.
Results: The sequential tam-ai strategy was less costly than the other strategies, but less effective than upfront ai and more effective than upfront tam. Upfront ai was more effective and less costly than upfront tam because of less breast cancer recurrence and differences in adverse events. In an exploratory analysis that included a sequential ai-tam strategy, ai-tam dominated based on small numerical differences unlikely to be clinically significant; that strategy was thus not used in the base-case analysis.
Conclusions: In postmenopausal women with er-positive early breast cancer, strategies using ais appear to provide more benefit than strategies using tam alone. Among the ai-containing strategies, sequential strategies using tam and an ai appear to provide benefits similar to those provided by upfront ai, but at a lower cost.
Keywords: Breast cancer; aromatase inhibitors; cost-effectiveness; tamoxifen.
Figures


Similar articles
-
Cost-utility of adjuvant hormone therapies with aromatase inhibitors in post-menopausal women with breast cancer: upfront anastrozole, sequential tamoxifen-exemestane and extended tamoxifen-letrozole.Breast. 2007 Jun;16(3):252-61. doi: 10.1016/j.breast.2006.12.002. Epub 2007 Jan 4. Breast. 2007. PMID: 17207623
-
Cost-effectiveness analysis of extended adjuvant endocrine therapy in the treatment of post-menopausal women with hormone receptor positive breast cancer.Breast Cancer Res Treat. 2014 Jun;145(2):267-79. doi: 10.1007/s10549-014-2950-6. Epub 2014 Apr 26. Breast Cancer Res Treat. 2014. PMID: 24771048
-
Cost-utility of adjuvant hormone therapies for breast cancer in post-menopausal women: sequential tamoxifen-exemestane and upfront anastrozole.Breast Cancer Res Treat. 2007 Mar;101(3):325-33. doi: 10.1007/s10549-006-9299-4. Epub 2006 Aug 2. Breast Cancer Res Treat. 2007. PMID: 16897433
-
Adjuvant therapy with aromatase inhibitors in postmenopausal, estrogen receptor- positive breast cancer patients: upfront or sequential?J BUON. 2009 Jul-Sep;14(3):375-9. J BUON. 2009. PMID: 19810126 Review.
-
Initial versus sequential adjuvant aromatase inhibitor therapy: a review of the current data.Curr Med Res Opin. 2006 Dec;22(12):2479-87. doi: 10.1185/030079906X154150. Curr Med Res Opin. 2006. PMID: 17257462 Review.
Cited by
-
Economic Evaluation of Letrozole for Early Breast Cancer in a Health Resource-Limited Setting.Biomed Res Int. 2018 Aug 2;2018:9282646. doi: 10.1155/2018/9282646. eCollection 2018. Biomed Res Int. 2018. PMID: 30155484 Free PMC article.
-
Isoforms of IDH in breast carcinoma: IDH2 as a potent prognostic factor associated with proliferation in estrogen-receptor positive cases.Breast Cancer. 2021 Jul;28(4):915-926. doi: 10.1007/s12282-021-01228-x. Epub 2021 Mar 12. Breast Cancer. 2021. PMID: 33713004
-
Systematic review of economic evaluations of aromatase inhibitors in estrogen receptor-positive breast cancer: quality evaluation.BMC Health Serv Res. 2023 Jun 26;23(1):689. doi: 10.1186/s12913-023-09432-5. BMC Health Serv Res. 2023. PMID: 37365615 Free PMC article.
-
The value-for-money of adjuvant aromatase inhibitors: time to put the debate to rest?Curr Oncol. 2015 Apr;22(2):77-9. doi: 10.3747/co.22.2579. Curr Oncol. 2015. PMID: 25908904 Free PMC article. No abstract available.
-
D-2-hydroxyglutarate dehydrogenase in breast carcinoma as a potent prognostic marker associated with proliferation.Histol Histopathol. 2021 Oct;36(10):1053-1062. doi: 10.14670/HH-18-362. Epub 2021 Jul 23. Histol Histopathol. 2021. PMID: 34296423
References
-
- Canadian Cancer Society’s Steering Committee on Cancer Statistics . Canadian Cancer Statistics 2012. Toronto, ON: Canadian Cancer Society; 2012.
-
- Forbes JF, Cuzick J, Buzdar A, Howell A, Tobias JS, Baum M, on behalf of the Arimidex, Tamoxifen, Alone or in Combination (atac) Trialists’ Group Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 100-month analysis of the atac trial. Lancet Oncol. 2008;9:45–53. doi: 10.1016/S1470-2045(07)70385-6. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

