Glucose administration after traumatic brain injury exerts some benefits and no adverse effects on behavioral and histological outcomes
- PMID: 25911580
- PMCID: PMC4457685
- DOI: 10.1016/j.brainres.2015.04.022
Glucose administration after traumatic brain injury exerts some benefits and no adverse effects on behavioral and histological outcomes
Abstract
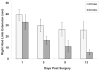
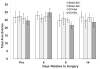
The impact of hyperglycemia after traumatic brain injury (TBI), and even the administration of glucose-containing solutions to head injured patients, remains controversial. In the current study adult male Sprague-Dawley rats were tested on behavioral tasks and then underwent surgery to induce sham injury or unilateral controlled cortical impact (CCI) injury followed by injections (i.p.) with either a 50% glucose solution (Glc; 2g/kg) or an equivalent volume of either 0.9% or 8% saline (Sal) at 0, 1, 3 and 6h post-injury. The type of saline treatment did not significantly affect any outcome measures, so these data were combined. Rats with CCI had significant deficits in beam-walking traversal time and rating scores (p's < 0.001 versus sham) that recovered over test sessions from 1 to 13 days post-injury (p's < 0.001), but these beam-walking deficits were not affected by Glc versus Sal treatments. Persistent post-CCI deficits in forelimb contraflexion scores and forelimb tactile placing ability were also not differentially affected by Glc or Sal treatments. However, deficits in latency to retract the right hind limb after limb extension were significantly attenuated in the CCI-Glc group (p < 0.05 versus CCI-Sal). Both CCI groups were significantly impaired in a plus maze test of spatial working memory on days 4, 9 and 14 post-surgery (p < 0.001 versus sham), and there was no effect of Glc versus Sal on this cognitive outcome measure. At 15 days post-surgery the loss of cortical tissue volume (p < 0.001 versus sham) was significantly less in the CCI-Glc group (30.0%; p < 0.05) compared to the CCI-Sal group (35.7%). Counts of surviving hippocampal hilar neurons revealed a significant (~40%) loss ipsilateral to CCI (p < 0.001 versus sham), but neuronal loss in the hippocampus was not different in the CCI-Sal and CCI-Glc groups. Taken together, these results indicate that an early elevation of blood glucose may improve some neurological outcomes and, importantly, the induction of hyperglycemia after isolated TBI did not adversely affect any sensorimotor, cognitive or histological outcomes.
Keywords: Beam-walking; Cognition; Controlled cortical impact; Contusion volume; Hippocampus; Hyperglycemia; Neurological exam; Rat; Spatial working memory.
Copyright © 2015 Elsevier B.V. All rights reserved.
Figures






Similar articles
-
Metabolic fate of glucose in rats with traumatic brain injury and pyruvate or glucose treatments: A NMR spectroscopy study.Neurochem Int. 2017 Jan;102:66-78. doi: 10.1016/j.neuint.2016.11.014. Epub 2016 Dec 3. Neurochem Int. 2017. PMID: 27919624 Free PMC article.
-
Attenuation of working memory and spatial acquisition deficits after a delayed and chronic bromocriptine treatment regimen in rats subjected to traumatic brain injury by controlled cortical impact.J Neurotrauma. 2002 Apr;19(4):415-25. doi: 10.1089/08977150252932370. J Neurotrauma. 2002. PMID: 11990348
-
Systemic administration of donepezil attenuates the efficacy of environmental enrichment on neurobehavioral outcome after experimental traumatic brain injury.Restor Neurol Neurosci. 2018;36(1):45-57. doi: 10.3233/RNN-170781. Restor Neurol Neurosci. 2018. PMID: 29439368
-
Criteria to define mild, moderate, and severe traumatic brain injury in the mouse controlled cortical impact model.Exp Neurol. 2018 Dec;310:48-57. doi: 10.1016/j.expneurol.2018.07.004. Epub 2018 Jul 12. Exp Neurol. 2018. PMID: 30017882 Review.
-
The Controlled Cortical Impact Model: Applications, Considerations for Researchers, and Future Directions.Front Neurol. 2016 Aug 17;7:134. doi: 10.3389/fneur.2016.00134. eCollection 2016. Front Neurol. 2016. PMID: 27582726 Free PMC article. Review.
Cited by
-
Metabolic fate of glucose in rats with traumatic brain injury and pyruvate or glucose treatments: A NMR spectroscopy study.Neurochem Int. 2017 Jan;102:66-78. doi: 10.1016/j.neuint.2016.11.014. Epub 2016 Dec 3. Neurochem Int. 2017. PMID: 27919624 Free PMC article.
-
Knockout of Cyclophilin-D Provides Partial Amelioration of Intrinsic and Synaptic Properties Altered by Mild Traumatic Brain Injury.Front Syst Neurosci. 2016 Jul 20;10:63. doi: 10.3389/fnsys.2016.00063. eCollection 2016. Front Syst Neurosci. 2016. PMID: 27489538 Free PMC article.
-
Chrysin attenuates traumatic brain injury-induced recognition memory decline, and anxiety/depression-like behaviors in rats: Insights into underlying mechanisms.Psychopharmacology (Berl). 2020 Jun;237(6):1607-1619. doi: 10.1007/s00213-020-05482-3. Epub 2020 Feb 22. Psychopharmacology (Berl). 2020. PMID: 32088834
-
Altering the Trajectory of Perfusion-Diffusion Deficits Using A BDNF Mimetic Acutely After TBI is Associated with Improved Functional Connectivity.Prog Neurobiol (Dover). 2023;10(1):10.60124/j.pneuro.2023.10.07. doi: 10.60124/j.pneuro.2023.10.07. Epub 2023 Sep 1. Prog Neurobiol (Dover). 2023. PMID: 38037566 Free PMC article.
-
Pyruvate treatment attenuates cerebral metabolic depression and neuronal loss after experimental traumatic brain injury.Brain Res. 2016 Jul 1;1642:270-277. doi: 10.1016/j.brainres.2016.04.005. Epub 2016 Apr 6. Brain Res. 2016. PMID: 27059390 Free PMC article.
References
-
- Alessandri B, Reinert M, Young HF, Bullock R. Low extracellular (ECF) glucose affects the neurochemical profile in severe head-injured patients. Acta Neurochir (Wein) Suppl. 2000;76:425–430. - PubMed
-
- Alessandri B, Schwandt E, Kamada Y, Nagata M, Heimann A, Kempski O. The neuroprotective effect of lactate is not due to improved glutamate uptake after controlled cortical impact in rats. J Neurotrauma. 2012;29:2181–2191. - PubMed
-
- Alves OL, Bullock R, Clausen T, Reinert M, Reeves TM. Concurrent monitoring of cerebral electrophysiology and metabolism after traumatic brain injury: an experimental and clinical study. J Neurotrauma. 2005;22:773–749. - PubMed
-
- Bartnik BL, Sutton RL, Fukushima M, Harris NG, Hovda DA, Lee SM. Upregulation of pentose phosphate pathway and preservation of tricarboxylic acid cycle flux after experimental brain injury. J Neurotrauma. 2005;22:1052–1065. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

