Randomized Controlled Trial of the Combined Effects of Web and Quitline Interventions for Smokeless Tobacco Cessation
- PMID: 25914872
- PMCID: PMC4405799
- DOI: 10.1016/j.invent.2015.02.005
Randomized Controlled Trial of the Combined Effects of Web and Quitline Interventions for Smokeless Tobacco Cessation
Abstract
Background: Use of smokeless tobacco (moist snuff and chewing tobacco) is a significant public health problem but smokeless tobacco users have few resources to help them quit. Web programs and telephone-based programs (Quitlines) have been shown to be effective for smoking cessation. We evaluate the effectiveness of a Web program, a Quitline, and the combination of the two for smokeless users recruited via the Web.
Objectives: To test whether offering both a Web and Quitline intervention for smokeless tobacco users results in significantly better long-term tobacco abstinence outcomes than offering either intervention alone; to test whether the offer of Web or Quitline results in better outcome than a self-help manual only Control condition; and to report the usage and satisfaction of the interventions when offered alone or combined.
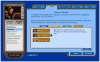
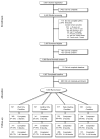
Methods: Smokeless tobacco users (N= 1,683) wanting to quit were recruited online and randomly offered one of four treatment conditions in a 2×2 design: Web Only, Quitline Only, Web + Quitline, and Control (printed self-help guide). Point-prevalence all tobacco abstinence was assessed at 3- and 6-months post enrollment.
Results: 69% of participants completed both the 3- and 6-month assessments. There was no significant additive or synergistic effect of combining the two interventions for Complete Case or the more rigorous Intent To Treat (ITT) analyses. Significant simple effects were detected, individually the interventions were more efficacious than the control in achieving repeated 7-day point prevalence all tobacco abstinence: Web (ITT, OR = 1.41, 95% CI = 1.03, 1.94, p = .033) and Quitline (ITT: OR = 1.54, 95% CI = 1.13, 2.11, p = .007). Participants were more likely to complete a Quitline call when offered only the Quitline intervention (OR = 0.71, 95% CI = .054, .093, p = .013), the number of website visits and duration did not differ when offered alone or in combination with Quitline. Rates of program helpfulness (p <.05) and satisfaction (p <.05) were higher for those offered both interventions versus offered only quitline.
Conclusion: Combining Web and Quitline interventions did not result in additive or synergistic effects, as have been found for smoking. Both interventions were more effective than a self-help control condition in helping motivated smokeless tobacco users quit tobacco. Intervention usage and satisfaction were related to the amount intervention content offered. Usage of the Quitline intervention decreased when offered in combination, though rates of helpfulness and recommendations were higher when offered in combination.
Trial registration: Clinicaltrials.gov NCT00820495; http://clinicaltrials.gov/ct2/show/NCT00820495.
Keywords: Web-based intervention; eHealth; quitline; smokeless tobacco; tobacco cessation.
Conflict of interest statement
None declared.
Figures
References
-
- Alguacil J, Silverman DT. Smokeless and other noncigarette tobacco use and pancreatic cancer: a case-control study based on direct interviews. Cancer Epidemiol Biomarkers Prev. 2004;13(1):55–58. - PubMed
-
- Baker TB, Piper ME, McCarthy DE, Bolt DM, Smith SS, Kim SY, Colby S, Conti D, Giovino GA, Hatsukami D, Hyland A, Krishnan-Sarin S, Niaura R, Perkins KA, Toll BA. Time to first cigarette in the morning as an index of ability to quit smoking: implications for nicotine dependence. Nicotine Tob Res. 2007;9(Suppl 4):S555–570. doi: 10.1080/14622200701673480. - DOI - PMC - PubMed
-
- Biener L, Abrams DB. The Contemplation Ladder: validation of a measure of readiness to consider smoking cessation. Health Psychol. 1991;10(5):360–365. - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous