Eltrombopag modulates reactive oxygen species and decreases acute myeloid leukemia cell survival
- PMID: 25915523
- PMCID: PMC4411049
- DOI: 10.1371/journal.pone.0126691
Eltrombopag modulates reactive oxygen species and decreases acute myeloid leukemia cell survival
Abstract
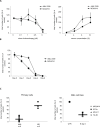
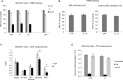
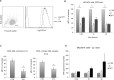
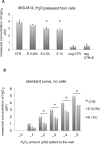
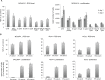
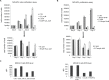
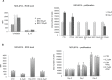
Previous studies have demonstrated that the small molecule thrombopoietin (TPO) mimetic, eltrombopag (E), induces apoptosis in acute myeloid leukemia (AML) cells. Here, we sought to define the mechanism of the anti-leukemic effect of eltrombopag. Our studies demonstrate that, at a concentration of 5 μM E in 2% serum, E induces apoptosis in leukemia cells by triggering PARP cleavage and activation of caspase cascades within 2-6 hours. The induction of apoptotic enzymes is critically dependent on drug concentration and the concentration of serum. This effect is not associated with an alteration in mitochondrial potential but is associated with a rapid decrease in a reactive oxygen species (ROS) in particular hydrogen peroxide (H2O2). Interestingly, E also decreases mitochondrial maximal and spare respiratory capacities suggesting an induced mitochondrial dysfunction that may not be readily apparent under basal conditions but becomes manifest only under stress. Co-treatment of MOLM14 AML cells with E plus Tempol or H2O2 provides a partial rescue of cell toxicity. Ferric ammonioum citrate (FAC) also antagonized the E induced toxicity, by inducing notable increase in ROS level. Overall, we propose that E dramatically decreases ROS levels leading to a disruption of AML intracellular metabolism and rapid cell death.
Conflict of interest statement
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

