Dynamic Virus-Bacterium Interactions in a Porcine Precision-Cut Lung Slice Coinfection Model: Swine Influenza Virus Paves the Way for Streptococcus suis Infection in a Two-Step Process
- PMID: 25916988
- PMCID: PMC4468551
- DOI: 10.1128/IAI.00171-15
Dynamic Virus-Bacterium Interactions in a Porcine Precision-Cut Lung Slice Coinfection Model: Swine Influenza Virus Paves the Way for Streptococcus suis Infection in a Two-Step Process
Abstract
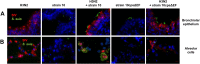
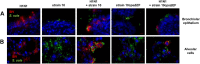
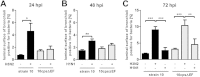
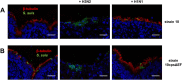
Swine influenza virus (SIV) and Streptococcus suis are common pathogens of the respiratory tract in pigs, with both being associated with pneumonia. The interactions of both pathogens and their contribution to copathogenesis are only poorly understood. In the present study, we established a porcine precision-cut lung slice (PCLS) coinfection model and analyzed the effects of a primary SIV infection on secondary infection by S. suis at different time points. We found that SIV promoted adherence, colonization, and invasion of S. suis in a two-step process. First, in the initial stages, these effects were dependent on bacterial encapsulation, as shown by selective adherence of encapsulated, but not unencapsulated, S. suis to SIV-infected cells. Second, at a later stage of infection, SIV promoted S. suis adherence and invasion of deeper tissues by damaging ciliated epithelial cells. This effect was seen with a highly virulent SIV subtype H3N2 strain but not with a low-virulence subtype H1N1 strain, and it was independent of the bacterial capsule, since an unencapsulated S. suis mutant behaved in a way similar to that of the encapsulated wild-type strain. In conclusion, the PCLS coinfection model established here revealed novel insights into the dynamic interactions between SIV and S. suis during infection of the respiratory tract. It showed that at least two different mechanisms contribute to the beneficial effects of SIV for S. suis, including capsule-mediated bacterial attachment to SIV-infected cells and capsule-independent effects involving virus-mediated damage of ciliated epithelial cells.
Copyright © 2015, American Society for Microbiology. All Rights Reserved.
Figures


References
-
- Fablet C, Marois C, Kuntz-Simon G, Rose N, Dorenlor V, Eono F, Eveno E, Jolly JP, Le Devendec L, Tocqueville V, Queguiner S, Gorin S, Kobisch M, Madec F. 2011. Longitudinal study of respiratory infection patterns of breeding sows in five farrow-to-finish herds. Vet Microbiol 147:329–339. doi:10.1016/j.vetmic.2010.07.005. - DOI - PMC - PubMed
-
- Meng F, Punyadarsaniya D, Uhlenbruck S, Hennig-Pauka I, Schwegmann-Wessels C, Ren X, Durrwald R, Herrler G. 2013. Replication characteristics of swine influenza viruses in precision-cut lung slices reflect the virulence properties of the viruses. Vet Res 44:110. doi:10.1186/1297-9716-44-110. - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

