SOX2 reprograms resident astrocytes into neural progenitors in the adult brain
- PMID: 25921813
- PMCID: PMC4437485
- DOI: 10.1016/j.stemcr.2015.03.006
SOX2 reprograms resident astrocytes into neural progenitors in the adult brain
Abstract
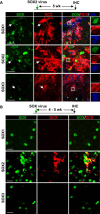
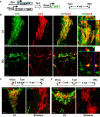
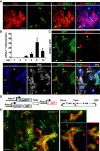
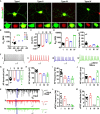
Glial cells can be in vivo reprogrammed into functional neurons in the adult CNS; however, the process by which this reprogramming occurs is unclear. Here, we show that a distinct cellular sequence is involved in SOX2-driven in situ conversion of adult astrocytes to neurons. This includes ASCL1(+) neural progenitors and DCX(+) adult neuroblasts (iANBs) as intermediates. Importantly, ASCL1 is required, but not sufficient, for the robust generation of iANBs in the adult striatum. These progenitor-derived iANBs predominantly give rise to calretinin(+) interneurons when supplied with neurotrophic factors or the small-molecule valproic acid. Patch-clamp recordings from the induced neurons reveal subtype heterogeneity, though all are functionally mature, fire repetitive action potentials, and receive synaptic inputs. Together, these results show that SOX2-mediated in vivo reprogramming of astrocytes to neurons passes through proliferative intermediate progenitors, which may be exploited for regenerative medicine.
Copyright © 2015 The Authors. Published by Elsevier Inc. All rights reserved.
Figures




References
-
- Abad M., Mosteiro L., Pantoja C., Cañamero M., Rayon T., Ors I., Graña O., Megías D., Domínguez O., Martínez D. Reprogramming in vivo produces teratomas and iPS cells with totipotency features. Nature. 2013;502:340–345. - PubMed
-
- Battiste J., Helms A.W., Kim E.J., Savage T.K., Lagace D.C., Mandyam C.D., Eisch A.J., Miyoshi G., Johnson J.E. Ascl1 defines sequentially generated lineage-restricted neuronal and oligodendrocyte precursor cells in the spinal cord. Development. 2007;134:285–293. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

