Regulation of Axolotl (Ambystoma mexicanum) Limb Blastema Cell Proliferation by Nerves and BMP2 in Organotypic Slice Culture
- PMID: 25923915
- PMCID: PMC4414535
- DOI: 10.1371/journal.pone.0123186
Regulation of Axolotl (Ambystoma mexicanum) Limb Blastema Cell Proliferation by Nerves and BMP2 in Organotypic Slice Culture
Abstract
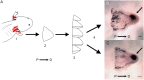
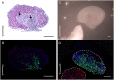
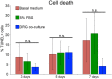
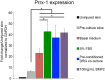
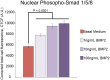
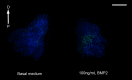
We have modified and optimized the technique of organotypic slice culture in order to study the mechanisms regulating growth and pattern formation in regenerating axolotl limb blastemas. Blastema cells maintain many of the behaviors that are characteristic of blastemas in vivo when cultured as slices in vitro, including rates of proliferation that are comparable to what has been reported in vivo. Because the blastema slices can be cultured in basal medium without fetal bovine serum, it was possible to test the response of blastema cells to signaling molecules present in serum, as well as those produced by nerves. We also were able to investigate the response of blastema cells to experimentally regulated changes in BMP signaling. Blastema cells responded to all of these signals by increasing the rate of proliferation and the level of expression of the blastema marker gene, Prrx-1. The organotypic slice culture model provides the opportunity to identify and characterize the spatial and temporal co-regulation of pathways in order to induce and enhance a regenerative response.
Conflict of interest statement
Figures


Similar articles
-
Nerve-induced ectopic limb blastemas in the Axolotl are equivalent to amputation-induced blastemas.Dev Biol. 2007 Dec 1;312(1):231-44. doi: 10.1016/j.ydbio.2007.09.021. Epub 2007 Sep 25. Dev Biol. 2007. PMID: 17959163
-
Blastema induction in aneurogenic state and Prrx-1 regulation by MMPs and FGFs in Ambystoma mexicanum limb regeneration.Dev Biol. 2011 Jul 15;355(2):263-74. doi: 10.1016/j.ydbio.2011.04.017. Epub 2011 Apr 22. Dev Biol. 2011. PMID: 21539833
-
Neurotrophic regulation of epidermal dedifferentiation during wound healing and limb regeneration in the axolotl (Ambystoma mexicanum).Dev Biol. 2008 Jul 15;319(2):321-35. doi: 10.1016/j.ydbio.2008.04.030. Epub 2008 May 3. Dev Biol. 2008. PMID: 18533144
-
The axolotl limb blastema: cellular and molecular mechanisms driving blastema formation and limb regeneration in tetrapods.Regeneration (Oxf). 2015 May 11;2(2):54-71. doi: 10.1002/reg2.32. eCollection 2015 Apr. Regeneration (Oxf). 2015. PMID: 27499868 Free PMC article. Review.
-
The role of peripheral nerves in urodele limb regeneration.Eur J Neurosci. 2011 Sep;34(6):908-16. doi: 10.1111/j.1460-9568.2011.07827.x. Eur J Neurosci. 2011. PMID: 21929624 Review.
Cited by
-
Rebuilding limbs, one cell at a time.Dev Dyn. 2022 Sep;251(9):1389-1403. doi: 10.1002/dvdy.463. Epub 2022 Mar 10. Dev Dyn. 2022. PMID: 35170828 Free PMC article.
-
Can neural signals override cellular decisions in the presence of DNA damage?DNA Repair (Amst). 2021 Jul;103:103127. doi: 10.1016/j.dnarep.2021.103127. Epub 2021 Apr 20. DNA Repair (Amst). 2021. PMID: 33990031 Free PMC article. Review.
-
Advancements to the Axolotl Model for Regeneration and Aging.Gerontology. 2020;66(3):212-222. doi: 10.1159/000504294. Epub 2019 Nov 28. Gerontology. 2020. PMID: 31779024 Free PMC article. Review.
-
Voltage-gated sodium channel scn8a is required for innervation and regeneration of amputated adult zebrafish fins.Proc Natl Acad Sci U S A. 2022 Jul 12;119(28):e2200342119. doi: 10.1073/pnas.2200342119. Epub 2022 Jul 6. Proc Natl Acad Sci U S A. 2022. PMID: 35867745 Free PMC article.
-
Neural control of growth and size in the axolotl limb regenerate.Elife. 2021 Nov 15;10:e68584. doi: 10.7554/eLife.68584. Elife. 2021. PMID: 34779399 Free PMC article.
References
-
- Globus M, Liversage RA (1975) In vitro studies of limb regeneration in adult Diemictylus viridescens: neural dependence of blastema cells for growth and differentiation. J Embryol Exp Morphol 33: 813–829. - PubMed
-
- Jabaily JA, Blue P, Singer M (1982) The Culturing of Dissociated Newt Forelimb Regenerate Cells. J Exp Zool 219: 67–73.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

