The Helicase Activity of Ribonuclease R Is Essential for Efficient Nuclease Activity
- PMID: 25931119
- PMCID: PMC4505480
- DOI: 10.1074/jbc.M115.650176
The Helicase Activity of Ribonuclease R Is Essential for Efficient Nuclease Activity
Abstract
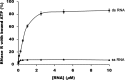
RNase R, which belongs to the RNB family of enzymes, is a 3' to 5' hydrolytic exoribonuclease able to digest highly structured RNA. It was previously reported that RNase R possesses an intrinsic helicase activity that is independent of its ribonuclease activity. However, the properties of this helicase activity and its relationship to the ribonuclease activity were not clear. Here, we show that helicase activity is dependent on ATP and have identified ATP-binding Walker A and Walker B motifs that are present in Escherichia coli RNase R and in 88% of mesophilic bacterial genera analyzed, but absent from thermophilic bacteria. We also show by mutational analysis that both of these motifs are required for helicase activity. Interestingly, the Walker A motif is located in the C-terminal region of RNase R, whereas the Walker B motif is in its N-terminal region implying that the two parts of the protein must come together to generate a functional ATP-binding site. Direct measurement of ATP binding confirmed that ATP binds only when double-stranded RNA is present. Detailed analysis of the helicase activity revealed that ATP hydrolysis is not required because both adenosine 5'-O-(thiotriphosphate) and adenosine 5'-(β,γ-imino)triphosphate can stimulate helicase activity, as can other nucleoside triphosphates. Although the nuclease activity of RNase R is not needed for its helicase activity, the helicase activity is important for effective nuclease activity against a dsRNA substrate, particularly at lower temperatures and with more stable duplexes. Moreover, competition experiments and mutational analysis revealed that the helicase activity utilizes the same catalytic channel as the nuclease activity. These findings indicate that the helicase activity plays an essential role in the catalytic efficiency of RNase R.
Keywords: ATP; Escherichia coli (E. coli); RNA degradation; RNA helicase; ribonuclease.
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures


Similar articles
-
Helicase Activity Plays a Crucial Role for RNase R Function in Vivo and for RNA Metabolism.J Biol Chem. 2016 Apr 29;291(18):9438-43. doi: 10.1074/jbc.C116.726091. Epub 2016 Mar 28. J Biol Chem. 2016. PMID: 27022019 Free PMC article.
-
How RNase R Degrades Structured RNA: ROLE OF THE HELICASE ACTIVITY AND THE S1 DOMAIN.J Biol Chem. 2016 Apr 8;291(15):7877-87. doi: 10.1074/jbc.M116.717991. Epub 2016 Feb 12. J Biol Chem. 2016. PMID: 26872969 Free PMC article.
-
The roles of individual domains of RNase R in substrate binding and exoribonuclease activity. The nuclease domain is sufficient for digestion of structured RNA.J Biol Chem. 2009 Jan 2;284(1):486-494. doi: 10.1074/jbc.M806468200. Epub 2008 Nov 11. J Biol Chem. 2009. PMID: 19004832 Free PMC article.
-
The Escherichia coli RNA degradosome: structure, function and relationship in other ribonucleolytic multienzyme complexes.Biochem Soc Trans. 2002 Apr;30(2):150-5. Biochem Soc Trans. 2002. PMID: 12035760 Review.
-
RNase II: the finer details of the Modus operandi of a molecular killer.RNA Biol. 2010 May-Jun;7(3):276-81. doi: 10.4161/rna.7.3.11490. Epub 2010 May 9. RNA Biol. 2010. PMID: 20484980 Review.
Cited by
-
Analysis of mRNA Decay Intermediates in Bacillus subtilis 3' Exoribonuclease and RNA Helicase Mutant Strains.mBio. 2022 Apr 26;13(2):e0040022. doi: 10.1128/mbio.00400-22. Epub 2022 Mar 21. mBio. 2022. PMID: 35311531 Free PMC article.
-
CircFISH: A Novel Method for the Simultaneous Imaging of Linear and Circular RNAs.Cancers (Basel). 2022 Jan 15;14(2):428. doi: 10.3390/cancers14020428. Cancers (Basel). 2022. PMID: 35053590 Free PMC article.
-
Structural basis of ribosomal 30S subunit degradation by RNase R.Nature. 2024 Feb;626(8001):1133-1140. doi: 10.1038/s41586-024-07027-6. Epub 2024 Feb 7. Nature. 2024. PMID: 38326618 Free PMC article.
-
Omnipresent Maxwell's demons orchestrate information management in living cells.Microb Biotechnol. 2019 Mar;12(2):210-242. doi: 10.1111/1751-7915.13378. Microb Biotechnol. 2019. PMID: 30806035 Free PMC article.
-
Direct observation of processive exoribonuclease motion using optical tweezers.Proc Natl Acad Sci U S A. 2015 Dec 8;112(49):15101-6. doi: 10.1073/pnas.1514028112. Epub 2015 Nov 23. Proc Natl Acad Sci U S A. 2015. PMID: 26598710 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

