Bone microenvironment signals in osteosarcoma development
- PMID: 25935149
- PMCID: PMC11113487
- DOI: 10.1007/s00018-015-1918-y
Bone microenvironment signals in osteosarcoma development
Abstract
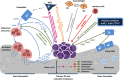
The bone is a complex connective tissue composed of many different cell types such as osteoblasts, osteoclasts, chondrocytes, mesenchymal stem/progenitor cells, hematopoietic cells and endothelial cells, among others. The interaction between them is finely balanced through the processes of bone formation and bone remodeling, which regulates the production and biological activity of many soluble factors and extracellular matrix components needed to maintain the bone homeostasis in terms of cell proliferation, differentiation and apoptosis. Osteosarcoma (OS) emerges in this complex environment as a result of poorly defined oncogenic events arising in osteogenic lineage precursors. Increasing evidence supports that similar to normal development, the bone microenvironment (BME) underlies OS initiation and progression. Here, we recapitulate the physiological processes that regulate bone homeostasis and review the current knowledge about how OS cells and BME communicate and interact, describing how these interactions affect OS cell growth, metastasis, cancer stem cell fate and therapy outcome.
Figures


References
-
- Kronenberg HM. Developmental regulation of the growth plate. Nature. 2003;423(6937):332–336. - PubMed
-
- Burdan F, Szumilo J, Korobowicz A, Farooquee R, Patel S, Patel A, Dave A, Szumilo M, Solecki M, Klepacz R, Dudka J. Morphology and physiology of the epiphyseal growth plate. Folia Histochem Cytobiol. 2009;47(1):5–16. - PubMed
-
- Wadayama B, Toguchida J, Shimizu T, Ishizaki K, Sasaki MS, Kotoura Y, Yamamuro T. Mutation spectrum of the retinoblastoma gene in osteosarcomas. Cancer Res. 1994;54(11):3042–3048. - PubMed
-
- Mutsaers AJ, Walkley CR. Cells of origin in osteosarcoma: mesenchymal stem cells or osteoblast committed cells? Bone. 2014;62:56–63. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

