Allosteric inhibition of factor XIa. Sulfated non-saccharide glycosaminoglycan mimetics as promising anticoagulants
- PMID: 25935648
- PMCID: PMC4526391
- DOI: 10.1016/j.thromres.2015.04.017
Allosteric inhibition of factor XIa. Sulfated non-saccharide glycosaminoglycan mimetics as promising anticoagulants
Abstract
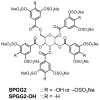
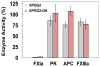
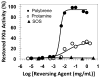
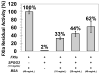
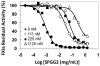
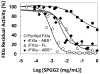
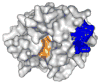
Recent development of sulfated non-saccharide glycosaminoglycan mimetics, especially sulfated pentagalloyl glucopyranoside (SPGG), as potent inhibitors of factor XIa (FXIa) (J. Med. Chem. 2013; 56:867-878 and J. Med. Chem. 2014; 57:4805-4818) has led to a strong possibility of developing a new line of factor XIa-based anticoagulants. In fact, SPGG represents the first synthetic, small molecule inhibitor that appears to bind in site remote from the active site. Considering that allosteric inhibition of FXIa is a new mechanism for developing a distinct line of anticoagulants, we have studied SPGG's interaction with FXIa with a goal of evaluating its pre-clinical relevance. Comparative inhibition studies with several glycosaminoglycans revealed the importance of SPGG's non-saccharide backbone. SPGG did not affect the activity of plasma kallikrein, activated protein C and factor XIIIa suggesting that SPGG-based anticoagulation is unlikely to affect other pathways connected with coagulation factors. SPGG's effect on APTT of citrated human plasma was also not dependent on antithrombin or heparin cofactor II. Interestingly, SPGG's anticoagulant potential was diminished by serum albumin as well as factor XI, while it could be reversed by protamine or polybrene, which implies possible avenues for developing antidote strategy. Studies with FXIa mutants indicated that SPGG engages Lys529, Arg530 and Arg532, but not Arg250, Lys252, Lys253 and Lys255. Finally, SPGG competes with unfractionated heparin, but not with polyphosphates and/or glycoprotein Ibα, for binding to FXIa. These studies enhance understanding on the first allosteric inhibitor of FXIa and highlight its value as a promising anticoagulant.
Copyright © 2015 Elsevier Ltd. All rights reserved.
Figures
Comment in
-
Medically-induced hemophilia C to treat thrombosis.Thromb Res. 2015 Aug;136(2):185-6. doi: 10.1016/j.thromres.2015.05.013. Epub 2015 May 22. Thromb Res. 2015. PMID: 26024825 No abstract available.
References
-
- Ageno W, Gallus AS, Wittkowsky A, Crowther M, Hylek EM, Palareti G American College of Chest Physicians. Oral anticoagulants: Antithrombotic therapy and prevention of thrombosis, 9th ed. : American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e44S–88S. - PMC - PubMed
-
- Crowther MA, Warkentin TE. Bleeding risk and the management of bleeding complications in patients undergoing anticoagulant therapy: focus on new anticoagulant agents. Blood. 2008;111:4871–4879. - PubMed
-
- Hirsh J, Anand SS, Halperin JL, Fuster V. Guide to anticoagulant therapy: Heparin: a statement for healthcare professionals from the American Heart Association. Circulation. 2001;103:2994–3018. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

