A cortical astrocyte subpopulation inhibits axon growth in vitro and in vivo
- PMID: 25936767
- PMCID: PMC4464481
- DOI: 10.3892/mmr.2015.3702
A cortical astrocyte subpopulation inhibits axon growth in vitro and in vivo
Abstract
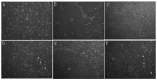
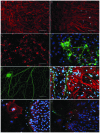
Astrocytes are the most heterogeneous and predominant glial cell type in the central nervous system. However, the functional significance of this heterogeneity remains to be elucidated. Following injury, damaged astrocytes inhibit axonal regeneration in vivo and in vitro. Cultured primary astrocytes are commonly considered good supportive substrates for neuron attachment and axon regeneration. However, it is not known whether different populations of cells in the heterogeneous astrocyte culture affect neuron behavior in the same way. In the present study, the effect of astrocyte heterogeneity on neuronal attachment and neurite outgrowth was examined using an in vitro and in vivo coculture system. In vitro, neonatal cortical astrocytes were co-cultured with purified dorsal root ganglia (DRG) neurons and astrocyte growth morphology, neuron attachment and neurite growth were evaluated. The results demonstrated that the heterogeneous astrocyte cells showed two different types of growth pattern, typical and atypical. Typical astrocytes were supportive to neuron attachment and neurite growth, which was consistent with previous studies, whereas atypical astrocytes inhibited neuron attachment and neurite growth. These inhibitory astrocytes exhibited a special growth pattern with various shapes and sizes, a high cell density, few oligodendrocytes on the top layer and occupied a smaller growth area compared with typical astrocytes. Neurites extended freely on typical supportive astrocyte populations, however, moved away when they reached atypical astrocyte growth pattern. Neurons growing on the atypical astrocyte pattern demonstrated minimal neurite outgrowth and these neurites had a dystrophic appearance, however, neuronal survival was unaffected. Immunocytochemistry studies demonstrated that these atypical inhibitory astrocytes were glial fibrillary acidic protein (GFAP) positive cells. The existence of inhibitory astrocyte subpopulations in normal astrocytes reflects the complexity of the function of astrocyte populations. In vivo, DRG neurons in grey matter did not show neurite growth, while DRG neurons survived and showed robust axon outgrowth along the corpus callosum. In conclusion, further studies on this new type of inhibitory astrocyte subpopulation may deepen our understanding of the complex biology of astrocytes.
Figures
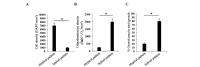


References
-
- Kettenmann H, Ransom BR, editors. Neuroglia. 2nd edition. Oxford University Press; New York: 2005. The concept of neuroglia: a historical perpspective; pp. 1–9.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

