Transcatheter embolization therapy in liver cancer: an update of clinical evidences
- PMID: 25937772
- PMCID: PMC4409973
- DOI: 10.3978/j.issn.1000-9604.2015.03.03
Transcatheter embolization therapy in liver cancer: an update of clinical evidences
Abstract
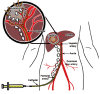
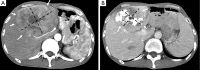
Transarterial chemoembolization (TACE) is a form of intra-arterial catheter-based chemotherapy that selectively delivers high doses of cytotoxic drug to the tumor bed combining with the effect of ischemic necrosis induced by arterial embolization. Chemoembolization and radioembolization are at the core of the treatment of liver hepatocellular carcinoma (HCC) patients who cannot receive potentially curative therapies such as transplantation, resection or percutaneous ablation. TACE for liver cancer has been proven to be useful in local tumor control, to prevent tumor progression, prolong patients' life and control patient symptoms. Recent evidence showed in patients with single-nodule HCC of 3 cm or smaller without vascular invasion, the 5-year overall survival (OS) with TACE was similar to that with hepatic resection and radiofrequency ablation. Although being used for decades, Lipiodol(®) (Lipiodol(®) Ultra Fluid(®), Guerbet, France) remains important as a tumor-seeking and radio-opaque drug delivery vector in interventional oncology. There have been efforts to improve the delivery of chemotherapeutic agents to tumors. Drug-eluting bead (DEB) is a relatively novel drug delivery embolization system which allows for fixed dosing and the ability to release the anticancer agents in a sustained manner. Three DEBs are available, i.e., Tandem(®) (CeloNova Biosciences Inc., USA), DC-Beads(®) (BTG, UK) and HepaSphere(®) (BioSphere Medical, Inc., USA). Transarterial radioembolization (TARE) technique has been developed, and proven to be efficient and safe in advanced liver cancers and those with vascular complications. Two types of radioembolization microspheres are available i.e., SIR-Spheres(®) (Sirtex Medical Limited, Australia) and TheraSphere(®) (BTG, UK). This review describes the basic procedure of TACE, properties and efficacy of some chemoembolization systems and radioembolization agents which are commercially available and/or currently under clinical evaluation. The key clinical trials of transcatheter arterial therapy for liver cancer are summarized.
Keywords: Lipiodol®; Transarterial; chemoembolization; drug-eluting particles; hepatocellular carcinoma (HCC); microspheres; radioembolization; yttrium-90.
Figures



References
-
- Ferlay J, Shin HR, Bray F, et al. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010;127:2893-917. - PubMed
-
- Kudo M, Izumi N, Kokudo N, et al. Management of hepatocellular carcinoma in Japan: Consensus-Based Clinical Practice Guidelines proposed by the Japan Society of Hepatology (JSH) 2010 updated version. Dig Dis 2011;29:339-64. - PubMed
-
- Bruix J, Castells A, Bosch J, et al. Surgical resection of hepatocellular carcinoma in cirrhotic patients: prognostic value of preoperative portal pressure. Gastroenterology 1996;111:1018-22. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
