Unraveling the pathogenesis of Hoyeraal-Hreidarsson syndrome, a complex telomere biology disorder
- PMID: 25940403
- PMCID: PMC4526362
- DOI: 10.1111/bjh.13442
Unraveling the pathogenesis of Hoyeraal-Hreidarsson syndrome, a complex telomere biology disorder
Abstract
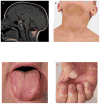
Hoyeraal-Hreidarsson (HH) syndrome is a multisystem genetic disorder characterized by very short telomeres and considered a clinically severe variant of dyskeratosis congenita. The main cause of mortality, usually in early childhood, is bone marrow failure. Mutations in several telomere biology genes have been reported to cause HH in about 60% of the HH patients, but the genetic defects in the rest of the patients are still unknown. Understanding the aetiology of HH and its diverse manifestations is challenging because of the complexity of telomere biology and the multiple telomeric and non-telomeric functions played by telomere-associated proteins in processes such as telomere replication, telomere protection, DNA damage response and ribosome and spliceosome assembly. Here we review the known clinical complications, molecular defects and germline mutations associated with HH, and elucidate possible mechanistic explanations and remaining questions in our understanding of the disease.
Keywords: Hoyeraal-Hreidarsson syndrome; cerebellar hypoplasia; dyskeratosis congenita; immunodeficiency; telomere.
© 2015 John Wiley & Sons Ltd.
Figures


References
-
- Aalfs CM, van den Berg H, Barth PG, Hennekam RC. The Hoyeraal-Hreidarsson syndrome: the fourth case of a separate entity with prenatal growth retardation, progressive pancytopenia and cerebellar hypoplasia. Eur J Pediatr. 1995;154:304–308. - PubMed
Publication types
MeSH terms
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

