Tumor indoleamine 2,3-dioxygenase (IDO) inhibits CD19-CAR T cells and is downregulated by lymphodepleting drugs
- PMID: 25940712
- PMCID: PMC4473118
- DOI: 10.1182/blood-2015-01-621474
Tumor indoleamine 2,3-dioxygenase (IDO) inhibits CD19-CAR T cells and is downregulated by lymphodepleting drugs
Abstract
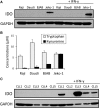
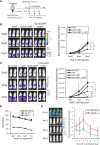
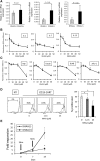
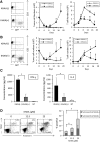
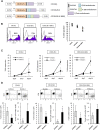
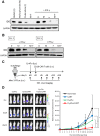
Although T cells expressing CD19-specific chimeric antigen receptors (CARs) are a promising new therapy for B-cell malignancies, objective responses are observed at lower frequencies in patients with lymphoma than in those with acute B-cell leukemia. We postulated that the tumor microenvironment suppresses CAR-expressing T cells (CARTs) through the activity of indoleamine 2,3-dioxygenase (IDO), an intracellular enzyme that converts tryptophan into metabolites that inhibit T -: cell activity. To investigate the effects of tumor IDO on CD19-CART therapy, we used a xenograft lymphoma model expressing IDO as a transgene. CD19-CARTs inhibited IDO-negative tumor growth but had no effect on IDO-positive tumors. An IDO inhibitor (1-methyl-tryptophan) restored IDO-positive tumor control. Moreover, tryptophan metabolites inhibited interleukin (IL)-2-, IL-7-, and IL-15-dependent expansion of CARTs; diminished their proliferation, cytotoxicity, and cytokine secretion in vitro in response to CD19 recognition; and increased their apoptosis. Inhibition of CD19-CARTs was not mitigated by the incorporation of costimulatory domains, such as 4-1BB, into the CD19-CAR. Finally, we found that fludarabine and cyclophosphamide, frequently used before CART administration, downregulated IDO expression in lymphoma cells and improved the antitumor activity of CD19-CART in vivo. Because tumor IDO inhibits CD19-CARTs, antagonizing this enzyme may benefit CD19-CART therapy.
© 2015 by The American Society of Hematology.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

