Minireview: Activin Signaling in Gonadotropes: What Does the FOX say… to the SMAD?
- PMID: 25942106
- PMCID: PMC5414707
- DOI: 10.1210/me.2015-1004
Minireview: Activin Signaling in Gonadotropes: What Does the FOX say… to the SMAD?
Abstract
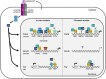
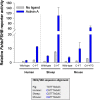
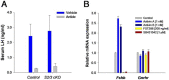
The activins were discovered and named based on their abilities to stimulate FSH secretion and FSHβ (Fshb) subunit expression by pituitary gonadotrope cells. According to subsequent in vitro observations, activins also stimulate the transcription of the GnRH receptor (Gnrhr) and the activin antagonist, follistatin (Fst). Thus, not only do activins stimulate FSH directly, they have the potential to regulate both FSH and LH indirectly by modulating gonadotrope sensitivity to hypothalamic GnRH. Moreover, activins may negatively regulate their own actions by stimulating the production of one of their principal antagonists. Here, we describe our current understanding of the mechanisms through which activins regulate Fshb, Gnrhr, and Fst transcription in vitro. The activin signaling molecules SMAD3 and SMAD4 appear to partner with the winged-helix/forkhead transcription factor, forkhead box L2 (FOXL2), to regulate expression of all 3 genes. However, in vivo data paint a different picture. Although conditional deletion of Foxl2 and/or Smad4 in murine gonadotropes produces impairments in FSH synthesis and secretion as well as in pituitary Fst expression, Gnrhr mRNA levels are either unperturbed or increased in these animals. Surprisingly, gonadotrope-specific deletion of Smad3 alone or with Smad2 does not impair FSH production or fertility; however, mice harboring these mutations may express a DNA binding-deficient, but otherwise functional, SMAD3 protein. Collectively, the available data firmly establish roles for FOXL2 and SMAD4 in Fshb and Fst expression in gonadotrope cells, whereas SMAD3's role requires further investigation. Gnrhr expression, in contrast, appears to be FOXL2, SMAD4, and, perhaps, activin independent in vivo.
Figures


References
-
- Ling N, Ying SY, Ueno N, et al. . A homodimer of the β-subunits of inhibin A stimulates the secretion of pituitary follicle stimulating hormone. Biochem Biophys Res Commun. 1986;138:1129–1137. - PubMed
-
- Ling N, Ying SY, Ueno N, et al. . Pituitary FSH is released by a heterodimer of the β-subunits from the two forms of inhibin. Nature. 1986;321:779–782. - PubMed
-
- Vale W, Rivier J, Vaughan J, et al. . Purification and characterization of an FSH releasing protein from porcine ovarian follicular fluid. Nature. 1986;321:776–779. - PubMed
-
- Rivier J, Spiess J, McClintock R, Vaughan J, Vale W. Purification and partial characterization of inhibin from porcine follicular fluid. Biochem Biophys Res Commun. 1985;133:120–127. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

