Differentiation of adipocytes and osteocytes from human adipose and placental mesenchymal stem cells
- PMID: 25945239
- PMCID: PMC4414992
Differentiation of adipocytes and osteocytes from human adipose and placental mesenchymal stem cells
Abstract
Objectives: Mesenchymal stem cells (MSC) can be isolated from adult tissues such as adipose tissue and other sources. Among these sources, adipose tissue (because of easy access) and placenta (due to its immunomodulatory properties, in addition to other useful properties), have attracted more attention in terms of research. The isolation and comparison of MSC from these two sources provides a proper source for clinical experimentation. The aim of this study was to compare the characteristics of MSC isolated from human adipose tissue and placenta.
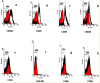
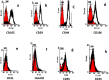
Materials and methods: Adipose and placental MSC were isolated from the subcutaneous adipose tissues of 10 healthy women (25 to 40 years) and from a fresh term placenta (n= 1), respectively. Stem cells were characterized and compared by flow cytometry using CD29, CD31, CD34, CD44, CD45, CD105, CD166 and HLA-DR markers. Osteocytes and adipocytes were differentiated from isolated human mesenchymal stem cells (HMSC).
Results: Adipose and placenta-derived MSC exhibited the same morphological features. ADSC differentiated faster than placenta; however, both were differentiated, taking up to 21 days for osteocyte and 14 days for adipocyte differentiation. About 90% of PLC-MSC and ADSC were positive for CD29, CD44, CD105, and CD166; and negative for CD31, CD34, CD45, and HLA-DR.
Conclusion: The two sources of stem cells showed similar surface markers, morphology and differentiation potential and because of their multipotency for differentiating to adipocytes and osteocytes, they can be applied as attractive sources of MSC for regenerative medicine.
Keywords: Adult stem cells; Differentiation; Fetal stem cells; Mesenchymal stem cells.
Conflict of interest statement
The authors confirm that there is no conflict of interest to disclose.
Figures

References
-
- Maqbool M, Vidyadaran S, George E, Ramasamy R. Human mesenchymal stem cells protect neutrophils from serum-deprived cell death. Cell Biol Int. 2011;35:1247–1251. - PubMed
-
- Semenov OV, Koestenbauer S, Riegel M, Zech N, Zimmermann R, Zisch AH, et al. Multipotent mesenchymal stem cells from human placenta: critical parameters for isolation and maintenance of stemness after isolation. Am J Obstet Gynecol. 2010;202:193 e1–193.e13. - PubMed
-
- Barlow S, Brooke G, Chatterjee K, Price G, Pelekanos R, Rossetti T, et al. Comparison of human placenta- and bone marrow-derived multipotent mesenchymal stem cells. Stem Cells Dev. 2008;17:1095–1107. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
