Increased expression of Solute carrier family 12 member 5 via gene amplification contributes to tumour progression and metastasis and associates with poor survival in colorectal cancer
- PMID: 25947013
- PMCID: PMC4819609
- DOI: 10.1136/gutjnl-2014-308257
Increased expression of Solute carrier family 12 member 5 via gene amplification contributes to tumour progression and metastasis and associates with poor survival in colorectal cancer
Abstract
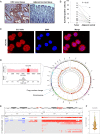
Objective: Using whole genome sequencing, we identified gene amplification of solute carrier family 12 member 5 (SLC12A5) located at 20q13.12 in colorectal cancer (CRC). We analysed its amplification, overexpression, biological effects and prognostic significance in CRC.
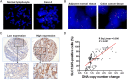
Design: SLC12A5 amplification status was evaluated by fluorescence in situ hybridisation (FISH). The effects of SLC12A5 re-expression or knockdown were determined in proliferation, apoptosis, invasion and metastasis assays. SLC12A5 target genes and related pathways were identified by reporter activity and cDNA microarray analyses. Clinical impact of SLC12A5 overexpression was assessed in 195 patients with CRC.
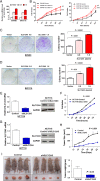
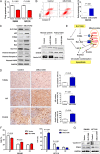
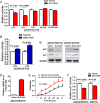
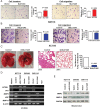
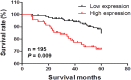
Results: Amplification of SLC12A5 was verified in 78 out of 191 (40.8%) patients with primary CRC by FISH, which was positively correlated with its protein overexpression (p<0.001). Biofunctional investigation of SLC12A5 revealed that SLC12A5 significantly increased cell proliferation, G1-S cell cycle transition, invasion/migration abilities, but suppressed apoptosis in vitro and promoted xenograft tumour growth as well as lung metastasis in vivo. The antiapoptosis effect by SLC12A5 was mediated through inhibiting apoptosis-inducing factor and endonuclease G-dependent apoptotic signalling pathway; and the pro-metastasis role was by regulating key elements of the matrix architecture, including matrix metallopeptidase and fibronectin. After a median follow-up of 50.16 months, multivariate analysis revealed that patients with SLC12A5 protein overexpression had a significant decrease in overall survival. Kaplan-Meier survival curves showed that SLC12A5 overexpression was significantly associated with shortened survival in patients with CRC.
Conclusions: SLC12A5 plays a pivotal oncogenic role in colorectal carcinogenesis; its overexpression is an independent prognostic factor of patients with CRC.
Keywords: COLORECTAL CANCER; GENE EXPRESSION; MOLECULAR ONCOLOGY; TUMOUR MARKERS.
Published by the BMJ Publishing Group Limited. For permission to use (where not already granted under a licence) please go to http://www.bmj.com/company/products-services/rights-and-licensing/
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
