Multiple sclerosis patient-derived CSF induces transcriptional changes in proliferating oligodendrocyte progenitors
- PMID: 25948622
- PMCID: PMC4628566
- DOI: 10.1177/1352458515573094
Multiple sclerosis patient-derived CSF induces transcriptional changes in proliferating oligodendrocyte progenitors
Abstract
Background: Cerebrospinal fluid (CSF) is in contact with brain parenchyma and ventricles, and its composition might influence the cellular physiology of oligodendrocyte progenitor cells (OPCs) thereby contributing to multiple sclerosis (MS) disease pathogenesis.
Objective: To identify the transcriptional changes that distinguish the transcriptional response induced in proliferating rat OPCs upon exposure to CSF from primary progressive multiple sclerosis (PPMS) or relapsing remitting multiple sclerosis (RRMS) patients and other neurological controls.
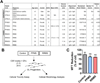
Methods: We performed gene microarray analysis of OPCs exposed to CSF from neurological controls, or definitive RRMS or PPMS disease course. Results were confirmed by quantitative reverse transcriptase polymerase chain reaction, immunocytochemistry and western blot of cultured cells, and validated in human brain specimens.
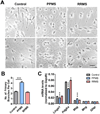
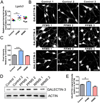
Results: We identified common and unique oligodendrocyte genes for each treatment group. Exposure to CSF from PPMS uniquely induced branching of cultured progenitors and related transcriptional changes, including upregulation (P<0.05) of the adhesion molecule GALECTIN-3/Lgals3, which was also detected at the protein level in brain specimens from PPMS patients. This pattern of gene expression was distinct from the transcriptional programme of oligodendrocyte differentiation during development.
Conclusions: Despite evidence of morphological differentiation induced by exposure to CSF of PPMS patients, the overall transcriptional response elicited in cultured OPCs was consistent with the activation of an aberrant transcriptional programme.
Keywords: Cerebrospinal fluid; differentiation; gene expression; oligodendrocyte progenitor cells.
© The Author(s), 2015.
Figures




References
-
- Franklin RJ, Ffrench-Constant C. Remyelination in the CNS: from biology to therapy. Nature reviews Neuroscience. 2008;9:839–855. - PubMed
-
- Hagemeier K, Bruck W, Kuhlmann T. Multiple sclerosis - remyelination failure as a cause of disease progression. Histology and histopathology. 2012;27:277–287. - PubMed
-
- Franklin RJ. Why does remyelination fail in multiple sclerosis? Nature reviews Neuroscience. 2002;3:705–714. - PubMed
-
- Rossi S, Motta C, Studer V, et al. Tumor necrosis factor is elevated in progressive multiple sclerosis and causes excitotoxic neurodegeneration. Multiple sclerosis. 2014;20:304–312. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

