Preserved Proteins from Extinct Bison latifrons Identified by Tandem Mass Spectrometry; Hydroxylysine Glycosides are a Common Feature of Ancient Collagen
- PMID: 25948757
- PMCID: PMC4587324
- DOI: 10.1074/mcp.M114.047787
Preserved Proteins from Extinct Bison latifrons Identified by Tandem Mass Spectrometry; Hydroxylysine Glycosides are a Common Feature of Ancient Collagen
Abstract
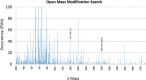
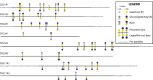
Bone samples from several vertebrates were collected from the Ziegler Reservoir fossil site, in Snowmass Village, Colorado, and processed for proteomics analysis. The specimens come from Pleistocene megafauna Bison latifrons, dating back ∼ 120,000 years. Proteomics analysis using a simplified sample preparation procedure and tandem mass spectrometry (MS/MS) was applied to obtain protein identifications. Several bioinformatics resources were used to obtain peptide identifications based on sequence homology to extant species with annotated genomes. With the exception of soil sample controls, all samples resulted in confident peptide identifications that mapped to type I collagen. In addition, we analyzed a specimen from the extinct B. latifrons that yielded peptide identifications mapping to over 33 bovine proteins. Our analysis resulted in extensive fibrillar collagen sequence coverage, including the identification of posttranslational modifications. Hydroxylysine glucosylgalactosylation, a modification thought to be involved in collagen fiber formation and bone mineralization, was identified for the first time in an ancient protein dataset. Meta-analysis of data from other studies indicates that this modification may be common in well-preserved prehistoric samples. Additional peptide sequences from extracellular matrix (ECM) and non-ECM proteins have also been identified for the first time in ancient tissue samples. These data provide a framework for analyzing ancient protein signatures in well-preserved fossil specimens, while also contributing novel insights into the molecular basis of organic matter preservation. As such, this analysis has unearthed common posttranslational modifications of collagen that may assist in its preservation over time. The data are available via ProteomeXchange with identifier PXD001827.
© 2015 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures



References
-
- Smejkal G. B., Schweitzer M. H. (2007) Will current technologies enable dinosaur proteomics? Expert Rev. Proteomics 4, 695–699 - PubMed
-
- Ostrom P. H., Schall M., Gandhi H., Shen T.-L., Hauschka P. V., Strahler J. R., Gage D. A. (2000) New strategies for characterizing ancient proteins using matrix-assisted laser desorption ionization mass spectrometry. Geochim. Cosmochim. Acta 64, 1043–1050
-
- Hofreiter M., Collins M., Stewart J. R. (2012) Ancient biomolecules in Quaternary palaeoecology. Quat. Sci. Rev. 33, 1–13
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

