BOLD fMRI of C-Fiber Mediated Nociceptive Processing in Mouse Brain in Response to Thermal Stimulation of the Forepaws
- PMID: 25950440
- PMCID: PMC4423852
- DOI: 10.1371/journal.pone.0126513
BOLD fMRI of C-Fiber Mediated Nociceptive Processing in Mouse Brain in Response to Thermal Stimulation of the Forepaws
Abstract
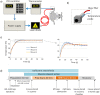
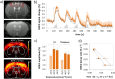
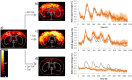
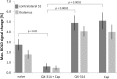
Functional magnetic resonance imaging (fMRI) in rodents enables non-invasive studies of brain function in response to peripheral input or at rest. In this study we describe a thermal stimulation paradigm using infrared laser diodes to apply noxious heat to the forepaw of mice in order to study nociceptive processing. Stimulation at 45 and 46°C led to robust BOLD signal changes in various brain structures including the somatosensory cortices and the thalamus. The BOLD signal amplitude scaled with the temperature applied but not with the area irradiated by the laser beam. To demonstrate the specificity of the paradigm for assessing nociceptive signaling we administered the quaternary lidocaine derivative QX-314 to the forepaws, which due to its positive charge cannot readily cross biological membranes. However, upon activation of TRPV1 channels following the administration of capsaicin the BOLD signal was largely abolished, indicative of a selective block of the C-fiber nociceptors due to QX-314 having entered the cells via the now open TRPV1 channels. This demonstrates that the cerebral BOLD response to thermal noxious paw stimulation is specifically mediated by C-fibers.
Conflict of interest statement
Figures
Similar articles
-
Increased blood oxygen level-dependent (BOLD) sensitivity in the mouse somatosensory cortex during electrical forepaw stimulation using a cryogenic radiofrequency probe.NMR Biomed. 2011 May;24(4):439-46. doi: 10.1002/nbm.1613. Epub 2010 Nov 30. NMR Biomed. 2011. PMID: 22945293
-
High field BOLD response to forepaw stimulation in the mouse.Neuroimage. 2010 Jun;51(2):704-12. doi: 10.1016/j.neuroimage.2010.02.083. Epub 2010 Mar 6. Neuroimage. 2010. PMID: 20211267
-
fMRI of pain processing in the brain: a within-animal comparative study of BOLD vs. CBV and noxious electrical vs. noxious mechanical stimulation in rat.Neuroimage. 2012 Jan 16;59(2):1168-79. doi: 10.1016/j.neuroimage.2011.08.002. Epub 2011 Aug 11. Neuroimage. 2012. PMID: 21856430
-
Mouse fMRI under ketamine and xylazine anesthesia: Robust contralateral somatosensory cortex activation in response to forepaw stimulation.Neuroimage. 2018 Aug 15;177:30-44. doi: 10.1016/j.neuroimage.2018.04.062. Epub 2018 May 4. Neuroimage. 2018. PMID: 29730495
-
Percept-related activity in the human somatosensory system: functional magnetic resonance imaging studies.Magn Reson Imaging. 2004 Dec;22(10):1539-48. doi: 10.1016/j.mri.2004.10.003. Magn Reson Imaging. 2004. PMID: 15707803 Review.
Cited by
-
Normothermic Mouse Functional MRI of Acute Focal Thermostimulation for Probing Nociception.Sci Rep. 2016 Jan 29;6:17230. doi: 10.1038/srep17230. Sci Rep. 2016. PMID: 26821826 Free PMC article.
-
Cell-specific Dyt1 ∆GAG knock-in to basal ganglia and cerebellum reveal differential effects on motor behavior and sensorimotor network function.Exp Neurol. 2023 Sep;367:114471. doi: 10.1016/j.expneurol.2023.114471. Epub 2023 Jun 14. Exp Neurol. 2023. PMID: 37321386 Free PMC article.
-
Functional optoacoustic neuro-tomography for scalable whole-brain monitoring of calcium indicators.Light Sci Appl. 2016 Dec 2;5(12):e16201. doi: 10.1038/lsa.2016.201. eCollection 2016 Dec. Light Sci Appl. 2016. PMID: 30167137 Free PMC article.
-
Generation of a whole-brain hemodynamic response function and sex-specific differences in cerebral processing of mechano-sensation in mice detected by BOLD fMRI.Front Neurosci. 2023 Aug 28;17:1187328. doi: 10.3389/fnins.2023.1187328. eCollection 2023. Front Neurosci. 2023. PMID: 37700753 Free PMC article.
-
Advancing Cardiovascular, Neurovascular, and Renal Magnetic Resonance Imaging in Small Rodents Using Cryogenic Radiofrequency Coil Technology.Front Pharmacol. 2015 Nov 12;6:255. doi: 10.3389/fphar.2015.00255. eCollection 2015. Front Pharmacol. 2015. PMID: 26617515 Free PMC article. Review.
References
-
- Ahrens ET, Dubowitz DJ. Peripheral somatosensory fMRI in mouse at 11.7 T. NMR Biomed. 2001; 14: 318–324. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

