Gastrointestinal perforation in metastatic colorectal cancer patients with peritoneal metastases receiving bevacizumab
- PMID: 25954110
- PMCID: PMC4419077
- DOI: 10.3748/wjg.v21.i17.5352
Gastrointestinal perforation in metastatic colorectal cancer patients with peritoneal metastases receiving bevacizumab
Abstract
Aim: To investigate the safety and efficacy of adding bevacizumab to first-line chemotherapy in metastatic colorectal cancer patients with peritoneal disease.
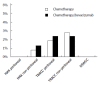
Methods: We compared rates of gastrointestinal perforation in patients with metastatic colorectal cancer and peritoneal disease receiving first-line chemotherapy with and without bevacizumab in three distinct cohorts: (1) the AGITG MAX trial (Phase III randomised clinical trial comparing capecitabine vs capecitabine and bevacizumab vs capecitabine, bevacizumab and mitomycinC); (2) the prospective Treatment of Recurrent and Advanced Colorectal Cancer (TRACC) registry (any first-line regimen ± bevacizumab); and (3) two cancer centres in New South Wales, Australia [Macarthur Cancer Therapy Centre and Liverpool Cancer Therapy Centre (NSWCC) from January 2005 to Decenber 2012, (any first-line regimen ± bevacizumab). For the AGITG MAX trial capecitabine was compared to the other two arms (capecitabine/bevacizumab and capecitabine/bevacizumab/mitomycinC). In the AGITG MAX trial and the TRACC registry rates of gastrointestinal perforation were also collected in patients who did not have peritoneal metastases. Secondary endpoints included progression-free survival, chemotherapy duration, and overall survival. Time-to-event outcomes were estimated using the Kaplan-Meier method and compared using the log-rank test.
Results: Eighty-four MAX, 179 TRACC and 69 NSWCC patients had peritoneal disease. There were no gastrointestinal perforations recorded in either the MAX subgroup or the NSWCC cohorts. Of the patients without peritoneal disease in the MAX trial, 4/300 (1.3%) in the bevacizumab arms had gastrointestinal perforations compared to 1/123 (0.8%) in the capecitabine alone arm. In the TRACC registry 3/126 (2.4%) patients who had received bevacizumab had a gastrointestinal perforation compared to 1/53 (1.9%) in the chemotherapy alone arm. In a further analysis of patients without peritoneal metastases in the TRACC registry, the rate of gastrointestinal perforations was 9/369 (2.4%) in the chemotherapy/bevacizumab group and 5/177 (2.8%) in the chemotherapy alone group. The addition of bevacizumab to chemotherapy was associated with improved progression-free survival in all three cohorts: MAX 6.9 m vs 4.9 m, HR = 0.64 (95%CI: 0.42-1.02); P = 0.063; TRACC 9.1 m vs 5.5 m, HR = 0.61 (95%CI: 0.37-0.86); P = 0.009; NSWCC 8.7 m vs 6.8 m, HR = 0.75 (95%CI: 0.43-1.32); P = 0.32. Chemotherapy duration was similar across the groups.
Conclusion: Patients with peritoneal disease do not appear to have an increased risk of gastrointestinal perforations when receiving first-line therapy with bevacizumab compared to systemic therapy alone.
Trial registration: ClinicalTrials.gov NCT00294359.
Keywords: Bevacizumab; Capecitabine; Colorectal neoplasms; Intestinal perforation; Peritoneal neoplasms.
Figures
Similar articles
-
Maintenance strategies after first-line oxaliplatin plus fluoropyrimidine plus bevacizumab for patients with metastatic colorectal cancer (AIO 0207): a randomised, non-inferiority, open-label, phase 3 trial.Lancet Oncol. 2015 Oct;16(13):1355-69. doi: 10.1016/S1470-2045(15)00042-X. Epub 2015 Sep 8. Lancet Oncol. 2015. PMID: 26361971 Clinical Trial.
-
Maintenance capecitabine and bevacizumab versus bevacizumab alone after initial first-line bevacizumab and docetaxel for patients with HER2-negative metastatic breast cancer (IMELDA): a randomised, open-label, phase 3 trial.Lancet Oncol. 2014 Nov;15(12):1351-60. doi: 10.1016/S1470-2045(14)70444-9. Epub 2014 Sep 28. Lancet Oncol. 2014. PMID: 25273343 Clinical Trial.
-
Capecitabine, bevacizumab, and mitomycin in first-line treatment of metastatic colorectal cancer: results of the Australasian Gastrointestinal Trials Group Randomized Phase III MAX Study.J Clin Oncol. 2010 Jul 1;28(19):3191-8. doi: 10.1200/JCO.2009.27.7723. Epub 2010 Jun 1. J Clin Oncol. 2010. PMID: 20516443 Clinical Trial.
-
Bevacizumab in combination with fluoropyrimidine-based chemotherapy for the first-line treatment of metastatic colorectal cancer.Health Technol Assess. 2010 Oct;14(Suppl. 2):47-53. doi: 10.3310/hta14suppl2/07. Health Technol Assess. 2010. PMID: 21047491 Review.
-
Bevacizumab combined with standard fluoropyrimidine-based chemotherapy regimens to treat colorectal cancer.Oncology. 2005;69 Suppl 3:17-24. doi: 10.1159/000088480. Epub 2005 Nov 21. Oncology. 2005. PMID: 16301832 Review.
Cited by
-
Chemotherapy plus bevacizumab versus chemotherapy plus cetuximab as first-line treatment for patients with metastatic colorectal cancer: Results of a registry-based cohort analysis.Medicine (Baltimore). 2016 Dec;95(51):e4531. doi: 10.1097/MD.0000000000004531. Medicine (Baltimore). 2016. PMID: 28002313 Free PMC article.
-
Effect of intraperitoneal cetuximab administration on colonic anastomosis and early postoperative adhesion formation in a rat model.Ulus Cerrahi Derg. 2015 Sep 1;32(3):157-61. doi: 10.5152/UCD.2016.3250. eCollection 2016. Ulus Cerrahi Derg. 2015. PMID: 27528807 Free PMC article.
References
-
- Lemmens VE, Klaver YL, Verwaal VJ, Rutten HJ, Coebergh JW, de Hingh IH. Predictors and survival of synchronous peritoneal carcinomatosis of colorectal origin: a population-based study. Int J Cancer. 2011;128:2717–2725. - PubMed
-
- Franko J, Shi Q, Goldman CD, Pockaj BA, Nelson GD, Goldberg RM, Pitot HC, Grothey A, Alberts SR, Sargent DJ. Treatment of colorectal peritoneal carcinomatosis with systemic chemotherapy: a pooled analysis of north central cancer treatment group phase III trials N9741 and N9841. J Clin Oncol. 2012;30:263–267. - PMC - PubMed
-
- Klaver YL, Simkens LH, Lemmens VE, Koopman M, Teerenstra S, Bleichrodt RP, de Hingh IH, Punt CJ. Outcomes of colorectal cancer patients with peritoneal carcinomatosis treated with chemotherapy with and without targeted therapy. Eur J Surg Oncol. 2012;38:617–623. - PubMed
-
- Kabbinavar FF, Flynn PJ, Kozloff M, Ashby MA, Sing A, Barr CE, Grothey A. Gastrointestinal perforation associated with bevacizumab use in metastatic colorectal cancer: results from a large treatment observational cohort study. Eur J Cancer. 2012;48:1126–1132. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical