Surfing along Filopodia: A Particle Transport Revealed by Molecular-Scale Fluctuation Analyses
- PMID: 25954870
- PMCID: PMC4423056
- DOI: 10.1016/j.bpj.2015.02.029
Surfing along Filopodia: A Particle Transport Revealed by Molecular-Scale Fluctuation Analyses
Abstract
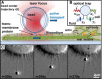
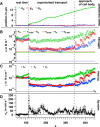
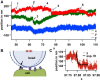
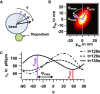
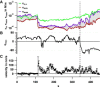
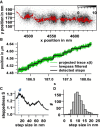
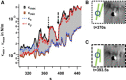
Filopodia perform cellular functions such as environmental sensing or cell motility, but they also grab for particles and withdraw them leading to an increased efficiency of phagocytic uptake. Remarkably, withdrawal of micron-sized particles is also possible without noticeable movements of the filopodia. Here, we demonstrate that polystyrene beads connected by optical tweezers to the ends of adherent filopodia of J774 macrophages, are transported discontinuously toward the cell body. After a typical resting time of 1-2 min, the cargo is moved with alternating velocities, force constants, and friction constants along the surface of the filopodia. This surfing-like behavior along the filopodium is recorded by feedback-controlled interferometric three-dimensional tracking of the bead motions at 10-100 kHz. We measured transport velocities of up to 120 nm/s and transport forces of ∼ 70 pN. Small changes in position, fluctuation width, and temporal correlation, which are invisible in conventional microscopy, indicate molecular reorganization of transport-relevant proteins in different phases of the entire transport process. A detailed analysis implicates a controlled particle transport with fingerprints of a nanoscale unbinding/binding behavior. The manipulation and analysis methods presented in our study may also be helpful in other fields of cellular biophysics.
Copyright © 2015 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Pulling, failing, and adaptive mechanotransduction of macrophage filopodia.Biophys J. 2022 Sep 6;121(17):3224-3241. doi: 10.1016/j.bpj.2022.07.028. Epub 2022 Aug 4. Biophys J. 2022. PMID: 35927956 Free PMC article.
-
Filopodia act as phagocytic tentacles and pull with discrete steps and a load-dependent velocity.Proc Natl Acad Sci U S A. 2007 Jul 10;104(28):11633-8. doi: 10.1073/pnas.0702449104. Epub 2007 Jul 9. Proc Natl Acad Sci U S A. 2007. PMID: 17620618 Free PMC article.
-
Filopodium retraction is controlled by adhesion to its tip.J Cell Sci. 2012 Nov 1;125(Pt 21):4999-5004. doi: 10.1242/jcs.104778. Epub 2012 Aug 16. J Cell Sci. 2012. PMID: 22899718 Free PMC article.
-
The making of filopodia.Curr Opin Cell Biol. 2006 Feb;18(1):18-25. doi: 10.1016/j.ceb.2005.11.002. Epub 2005 Dec 6. Curr Opin Cell Biol. 2006. PMID: 16337369 Review.
-
Filopodia: the fingers that do the walking.Sci STKE. 2007 Aug 21;2007(400):re5. doi: 10.1126/stke.4002007re5. Sci STKE. 2007. PMID: 17712139 Review.
Cited by
-
Pulling, failing, and adaptive mechanotransduction of macrophage filopodia.Biophys J. 2022 Sep 6;121(17):3224-3241. doi: 10.1016/j.bpj.2022.07.028. Epub 2022 Aug 4. Biophys J. 2022. PMID: 35927956 Free PMC article.
-
Involvement of two uptake mechanisms of gold and iron oxide nanoparticles in a co-exposure scenario using mouse macrophages.Beilstein J Nanotechnol. 2017 Nov 14;8:2396-2409. doi: 10.3762/bjnano.8.239. eCollection 2017. Beilstein J Nanotechnol. 2017. PMID: 29234575 Free PMC article.
-
Miniature scanning light-sheet illumination implemented in a conventional microscope.Biomed Opt Express. 2018 Aug 14;9(9):4263-4274. doi: 10.1364/BOE.9.004263. eCollection 2018 Sep 1. Biomed Opt Express. 2018. PMID: 30615716 Free PMC article.
-
Real Time Analysis of Bovine Viral Diarrhea Virus (BVDV) Infection and Its Dependence on Bovine CD46.Viruses. 2020 Jan 17;12(1):116. doi: 10.3390/v12010116. Viruses. 2020. PMID: 31963539 Free PMC article.
-
Bacterial Extracellular Vesicles Exploit Filopodial Surfing and Retraction Mechanisms to Reach the Host Cell Body in an Actin-Dependent Manner.J Extracell Vesicles. 2025 Jun;14(6):e70107. doi: 10.1002/jev2.70107. J Extracell Vesicles. 2025. PMID: 40545962 Free PMC article.
References
-
- Sheetz M.P., Wayne D.B., Pearlman A.L. Views and reviews extension of filopodia by motor-dependent actin assembly. Cell Motil. Cytoskeleton. 1992;169:160–169. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

